Exploring The Diversity Of Matter By Its Chemical Composition
In this article, the following are the objectives that will be discussed.
- Understand that every substance consists of chemical components.
- Understand what an element is.
- Understand how to use the Periodic Table.
- Using the Periodic table, write the chemical symbols of elements.
Chemical properties of Substances
Introduction
Different substances have different physical characteristics, which serve different functions. The different substances have different physical properties due to their chemical composition.
Chemical Composition
Chemical composition is the chemical components which make up a substance. Hence, matter can be classified as elements and non-elements, based on their chemical composition. Non-elements can be further classified into compounds and mixtures.
Elements
An element is the simplest form of matter. An element is a substance that cannot be broken down into two or more simpler substances by chemical methods.
Periodic Table
The Periodic Table is an arrangement of all the elements, in accordance with their increasing atomic number (proton number).
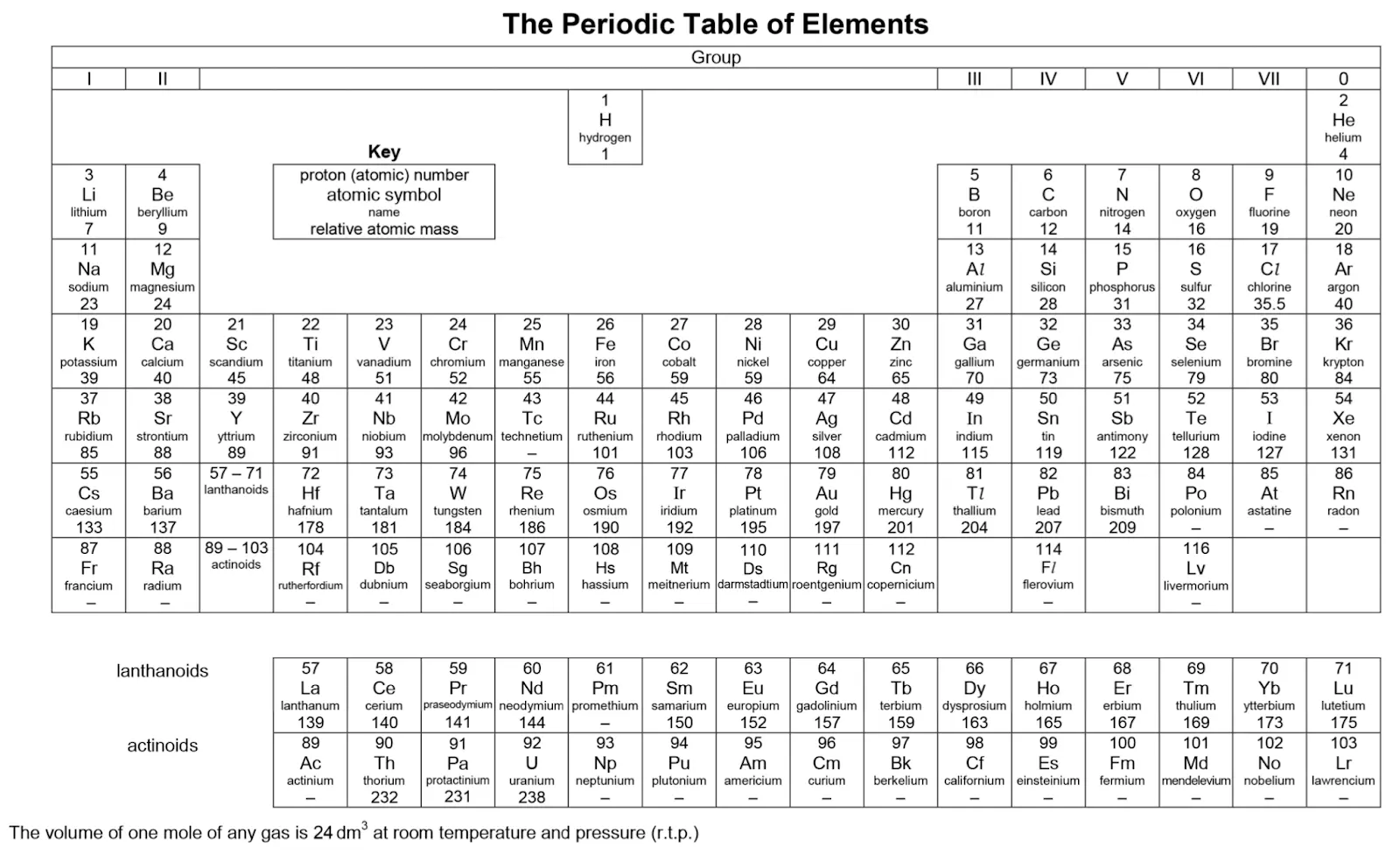
Image Credit: SEAB
Group
A group consists of elements which are found in the same vertical column. Elements which are arranged in the same column belong to the same group.
Image Credit: SEAB
Groups start from Group 1 to Group 18.
Elements which are in the same group have similar chemical properties, as they have the same number of valence electrons.
Period
Elements which are arranged in the same row belong to the same period.
Image Credit: SEAB
Moving from the left to the right of a particular period, the properties of elements gradually change from those of metals to those of non-metals.
Metallic and Non-Metallic Elements
Elements can be classified into metals and non-metals.
Here are some properties of metals:
- good conductor of heat and electricity
- high melting point and boiling point
- high density
- malleable and ductile
Here are some properties of non-metals:
- poor conductor of heat and electricity
- low melting point and boiling point
- dull and brittle
Symbols of Elements
- The elements can be represented as symbols.
- The symbols of elements consist of one or two letters.
- If the symbol of the element consists of only one letter, it must be written with an upper case.
Examples:
H (hydrogen), O (oxygen), N (nitrogen)
- If the symbol of the element consists of two letters, the first letter will be in upper case and the second one will be in lower case.
Examples:
Ne (neon), Li (lithium), Cl (chlorine)
Revision Questions
Here are some questions for us to look into on the objectives of this article.
Question 1:
Write the chemical symbol of each element below:
- iron
- carbon
- fluorine
- silver
- bismuth
- mercury
- magnesium
- chlorine
- aluminium
- cobalt
- barium
- manganese
Solution:
- iron → Fe
- carbon → C
- fluorine → F
- silver → Ag
- bismuth → Bi
- mercury → Hg
- magnesium → Mg
- chlorine → Cl
- aluminum → Al
- cobalt → Co
- barium → Ba
- manganese → Mn
Question 2:
Which of the following describes the elements correctly?
- They can be broken down into simpler forms by chemical means.
- They are not pure substances.
- They can be classified into two groups, metals and non-metals.
- There are more non-metallic elements than metallic ones.
Solution:
(C) They can be classified into two groups, metals and non-metals.
Explanation:
Elements are pure substances that cannot be broken down into simpler forms by chemical means. Based on the Periodic Table, there are more metallic elements than non-metallic elements.
Question 3:
Which of the following correctly classifies the elements that belong to the same column in the Periodic Table?
- chemical properties
- group
- physical Properties
- period
Solution:
(B) group
Explanation:
Elements in the same horizontal column belong to the same group.
Question 4:
Which of the following correctly classifies the elements that belong to the same row in the Periodic Table?
- chemical Properties
- group
- physical Properties
- period
Solution:
(D) period
Explanation:
Elements in the same row belong to the same period.
Question 5:
Which of the following properties describes the elements that belong to the same group of the Periodic Table?
- identical chemical properties
- identical physical properties
- similar chemical properties
- similar physical properties
Solution:
(C) similar chemical properties
Explanation:
Elements that belong to the same group of the Periodic Table have similar chemical properties. The physical properties of elements in the same group change gradually as one moves down the group.
Question 6:
Using the Periodic Table, which of the following elements is a metal?
- chlorine
- nitrogen
- sodium
- xenon
Solution:
(C) sodium
Explanation:
Chlorine, nitrogen, and xenon are non-metals.
Question 7:
Using the Periodic Table, which of the following elements is a metal?
- barium
- bromine
- phosphorus
- radon
Solution:
(A) barium
Explanation:
Bromine, phosphorus, and radon are non-metals.
Question 8:
Using the Periodic Table, which of the following elements is a non-metal?
- aluminium
- carbon
- magnesium
- titanium
Solution:
(B) carbon
Explanation:
Aluminium, magnesium, and titanium are metals.
Question 9:
Using the Periodic Table, which of the following symbols represents copper?
- Ca
- Ci
- Cu
- Co
Solution:
(C) Cu
Explanation:
Ca is the symbol of calcium.
Ci does not represent any elements on the Periodic Table.
Co is the symbol of cobalt.
| Continue Learning | |
|---|---|
| Model Of Cells - The Basic Unit Of Life | Solutions And Suspensions |
| Model Of Matter - Atoms And Molecules | Exploring The Diversity Of Matter By Its Chemical Composition |
| Exploring Diversity Of Matter By Its Physical Properties | Exploring Diversity Of Matter Using Separation Techniques |
| Model of Matter - The Particulate Nature Of Matter | Ray Model Of Light |
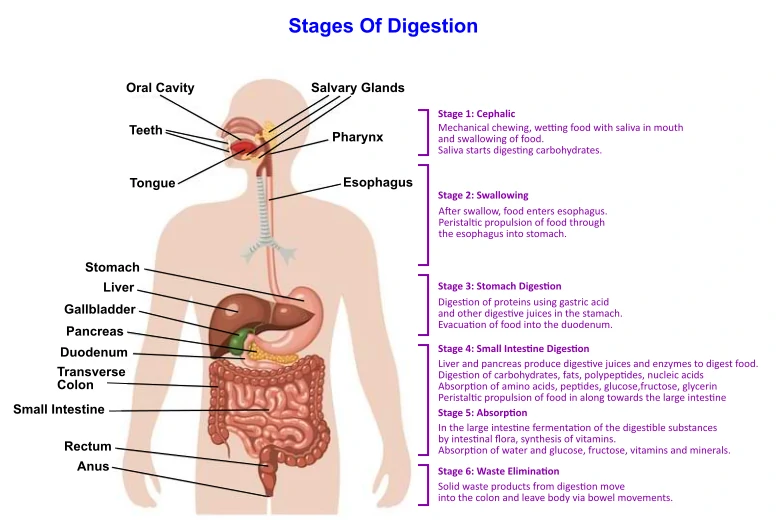
| Human Digestive System | |
 SG
SG  VN
VN