Salts
In this article, we will learn about the topic of Salts. We will focus on the following learning outcomes:
- Define a salt.
- Describe the general rules for the solubility of common salts in water.
- Describe the technique used and starting materials required in the preparation of insoluble salts.
- Write the balanced chemical and ionic equation, with state symbols, for the preparation of insoluble salts.
This article is specifically written to meet the requirements of the Upper Secondary Chemistry syllabus in Singapore.
What is a Salt?
Salts are ionic compounds, which contain both a cation and an anion.
What are the reactions that will produce salt?
- acid + metal ⟶ salt + hydrogen
- acid + carbonate ⟶ salt + water + carbon dioxide
- acid + base/alkali ⟶ salt + water
The cation in a salt is contributed by the reactant containing the metal in a reaction. The anion in a salt is contributed by the acid in a reaction.
Salts are ionic compounds formed by replacing one or more hydrogen ions of an acid with a metallic ion or an ammonium ion (NH4+).
Take a look at the following reaction between aqueous sodium hydroxide and dilute hydrochloric acid.
NaOH (aq) + HCl (aq) ⟶ NaCl (aq)+ H2O (l)
In the above chemical reaction, the metallic ion, Na⁺, from NaOH replaces the hydrogen ion of the acid, HCl, to give the salt sodium chloride, NaCl.
Take a look at another reaction, between magnesium carbonate solution and dilute sulfuric acid.
MgCO3 (aq) + H2SO4 (aq) ⟶ MgSO4 (aq) + H2O (l) + CO2 (g)
In the above chemical reaction, the metallic ion, Mg²⁺, from MgCO3 replaces the hydrogen ions of the acid, H2SO4, to give the salt magnesium sulfate, MgSO4.
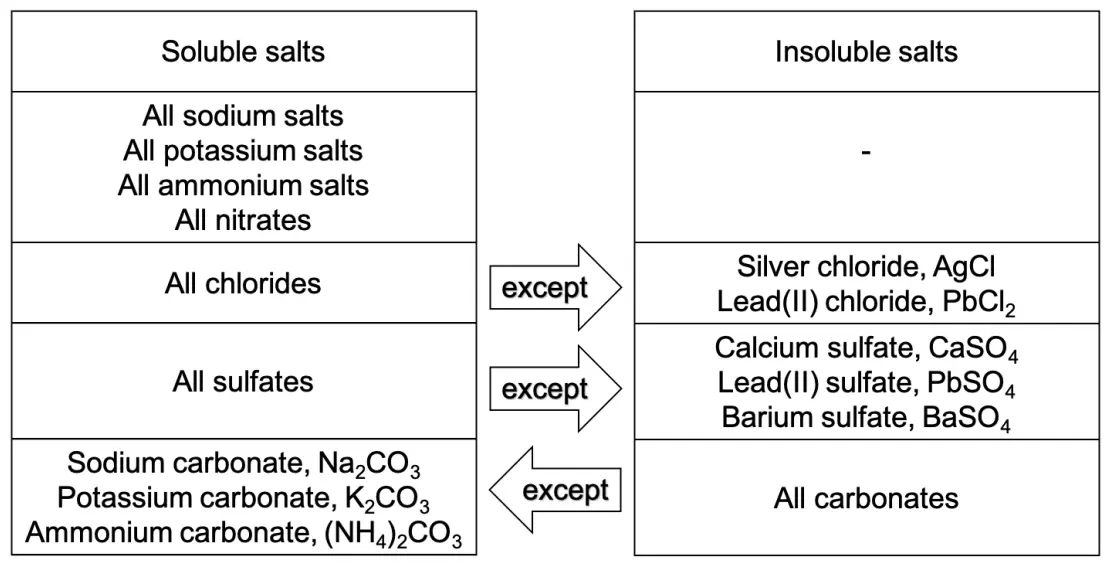
Solubility Table
Even though salts are ionic compounds, not all salts are soluble in water. The chart below shows the solubilities of the common salts in water.
Considerations
Before preparing a salt, you need to consider two factors:
- Is the salt soluble or insoluble in water?
- Are the starting materials (reactants) soluble in water?
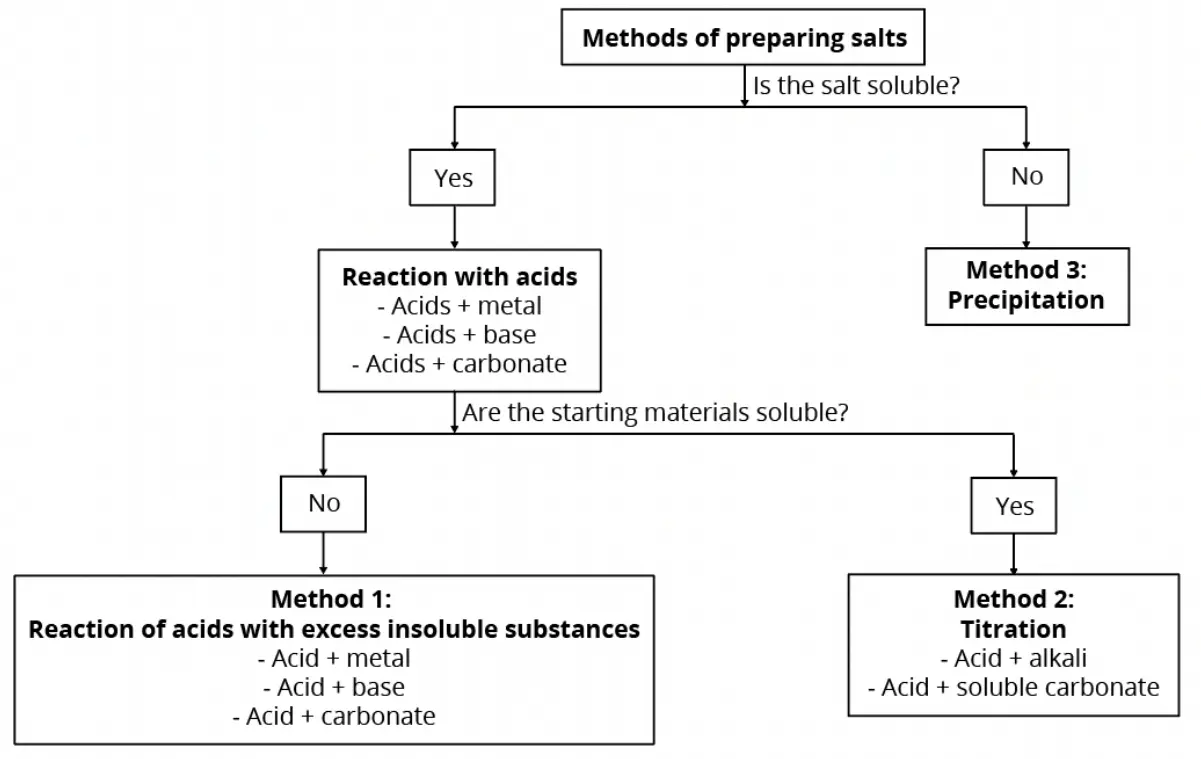
Which method should I choose?
The flowchart below summarises the methods of preparing salts in the laboratory.
In this article, we will focus first on the method to prepare insoluble salts.
Precipitation
What kinds of salts does precipitation prepare?
Precipitation is the method for preparing insoluble salts.
Examples of insoluble salts:
- silver chloride
- lead(II) chloride
- calcium sulfate
- lead(II) sulfate
- barium sulfate
Precipitation involves mixing two solutions to form an insoluble solid that can be separated from the reaction mixture.
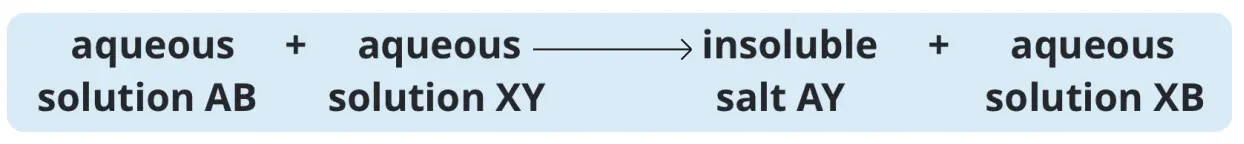
Each of the two aqueous solutions that you mix must contain either the cation or the anion of the insoluble salt to be prepared.
For Example:
Insoluble barium sulfate, BaSO4, can be prepared by using barium nitrate solution and dilute sulfuric acid or any soluble sulfate.
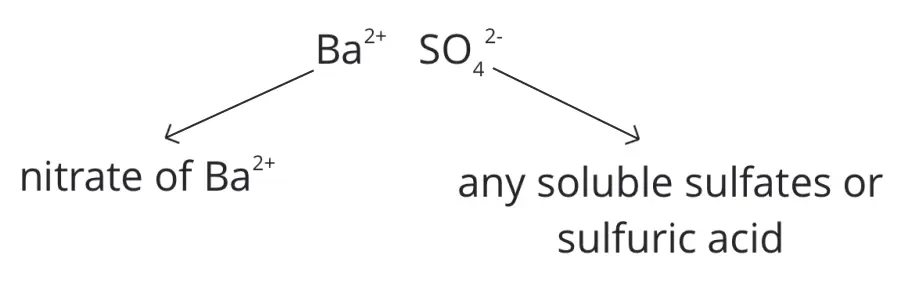
Here, we see that one of the aqueous solutions contains the barium ion while the other aqueous solution contains the sulfate ion.
Assuming that we mix barium nitrate solution and sodium sulfate solution to form the insoluble barium sulfate salt, the following are the word and chemical equations of this precipitation reaction.
Word Equation:
barium nitrate + sodium sulfate ⟶ barium sulfate + sodium nitrate
Chemical Equation:
Ba(NO3)2 (aq) + Na2SO4 (aq) ⟶ BaSO4 (s) + 2NaNO3 (aq)
Question 1:
Which salt would not be prepared by using precipitation method?
- barium carbonate
- lead(II) sulfate
- magnesium carbonate
- silver nitrate
Solution:
(D) silver nitrate
Explanation:
All nitrates are soluble in water. Precipitation is used to prepare insoluble salts. Therefore, silver nitrate cannot be prepared using the precipitation method.
Barium carbonate, lead(II) sulfate and magnesium carbonate are all insoluble salts, that can be prepared using the precipitation method.
Question 2:
Which three salts can all be prepared by precipitation?
- barium sulfate, lead(II) sulfate and calcium sulfate
- calcium sulfate, calcium carbonate and calcium chloride
- silver chloride, silver nitrate and silver sulfate
- zinc carbonate, potassium carbonate and iron(III) carbonate
Solution:
(A) barium sulfate, lead(II) sulfate and calcium sulfate
Explanation:
Precipitation is used to prepare insoluble salts.
All sulfates are soluble in water except barium sulfate, lead(II) sulfate and calcium sulfate. Therefore, barium sulfate, lead(II) sulfate and calcium sulfate can be prepared by precipitation.
Steps to prepare Insoluble Salts
To prepare barium sulfate:
Ba(NO3)2 (aq) + Na2SO4 (aq) ⟶ BaSO4 (s) + 2NaNO3 (aq)
- Pour about 25 cm3 of barium nitrate solution into a beaker. Add sodium sulfate solution (in excess) and stir until no more precipitate forms.
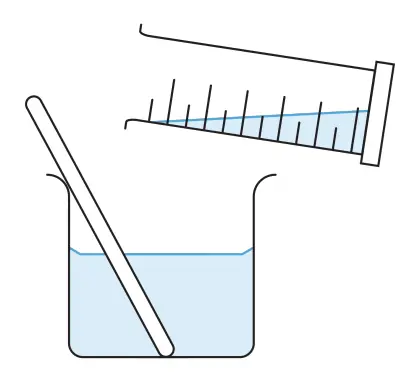
- Filter to collect the precipitate (barium sulfate) as the residue.
* The filtrate will contain sodium nitrate solution as well as the excess sodium sulfate solution.*
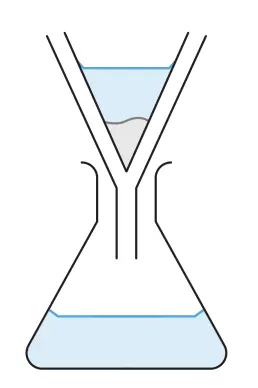
- Wash the precipitate with a little distilled water to remove impurities. (The impurities can be excess sodium sulfate solution or sodium nitrate solution.)
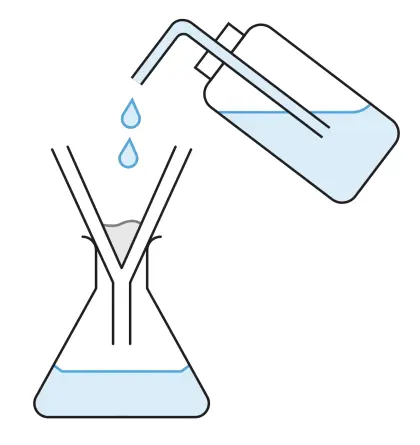
- Allow the precipitate to dry on a piece of filter paper.
Ionic Equation for the Preparation of Insoluble Salts
Let us practise writing the ionic equation for the preparation of insoluble barium sulfate from the reaction between barium nitrate solution and sodium sulfate solution.
Step 1: Write the balanced chemical equation, including the state symbols, for the reaction.
Ba(NO3)2 (aq) + Na2SO4 (aq) ⟶ BaSO4 (s) + 2NaNO3 (aq)
Step 2: Rewriting the equation in terms of ions for substances in the aqueous state.
Ba(NO3)2, Na2SO4 and 2NaNO3 are soluble in water. They exist as ions in solution. When we rewrite the chemical equation in terms of ions, we get:
Ba2+ (aq) + 2NO3− (aq) + 2Na+ (aq) + SO42− (aq) → BaSO4 (s) + 2Na+ (aq) + 2NO3− (aq)
Step 3: Cancel the spectator ions.
Spectator ions are ions that have not taken part in any chemical reaction. They have not changed their states before and after the reaction.
- Na+ (aq) and NO3− (aq) ions have not changed their states during the reaction. They are still ions in solution at the end of the reaction. Hence, Na+ (aq) and NO3− (aq) are spectator ions, and they will be left out of the equation.
Step 4: Write the ionic equation.
So, the final ionic equation will be:
Ba2+ (aq) + SO42- (aq) ⟶ BaSO4 (s)
Question 3:
Which of the following equations best represents the method to prepare an insoluble salt?
- Pb (s) + 2HCl (aq) ⟶ PbCl2 (s) + H2 (g)
- Pb(NO3)2 (aq) + 2HCl (aq) ⟶ PbCl2 (s) + 2HNO3 (aq)
- PbO (s) + 2HCl (aq) ⟶ PbCl2 (s) + H2O (I)
- PbSO4 (s) + HCl (aq) ⟶ PbCl2 (s) + H2SO4 (aq)
Solution:
(B) Pb(NO3)2 (aq) + 2HCl (aq) ⟶ PbCl2 (s) + 2HNO3 (aq)
Explanation:
When preparing an insoluble salt using the precipitation method, two aqueous solutions are needed. One aqueous solution should contain cation and the other aqueous solution should contain anion of the insoluble salt to be prepared.
To prepare the insoluble salt, lead(II) chloride, PbCl2 , we can mix lead(II) nitrate solution, Pb(NO3)2, with dilute hydrochloric acid, HCl. After mixing the two aqueous solutions, we can filter to collect the insoluble salt as the residue.
Conclusion
In the article, we learned about the general rules for the solubility of common salts in water. We also learned about the different methods involved in preparing salts in the laboratory. We focused on the steps involved in the precipitation method to prepare insoluble salts and how to construct the ionic equation.
 SG
SG  VN
VN