Separation And Purification
In this article, we will cover the following learning outcomes:
- Understand the difference between a pure substance and a mixture
- Identifying substances and their purity, given their melting and boiling points
- Describe methods of separating and purifying mixtures of a solid and a liquid
This article is written to meet the requirements of Secondary 3 Chemistry students.
Pure Substance
What is a pure substance?
A pure substance is made up of one single element or compound. It is not mixed with any other substances.
Examples:
- Distilled water (H2O) is a pure substance. It is a compound made up of hydrogen and oxygen atoms chemically combined together.
- Diamond is a pure substance because it is made of carbon atoms that are all chemically combined together. Diamond is an element.
- Common salt (also known as sodium chloride, NaCl) is a pure substance. It is a compound made up of sodium and chlorine atoms chemically combined together.
|
Image Credit: unsplash - Tahlia Doyle |
Image Credit: unsplash - Jason Tuinstra |
How do we test the purity of a substance?
The two methods we can use to test the purity of the substance are:
- Measure the melting and boiling point of a substance
- Perform chromatography
Melting and boiling point of a substance
-
Pure Substance
- Melting Point: A pure substance has a fixed melting point.
- Boiling Point: A pure substance has a fixed boiling point.
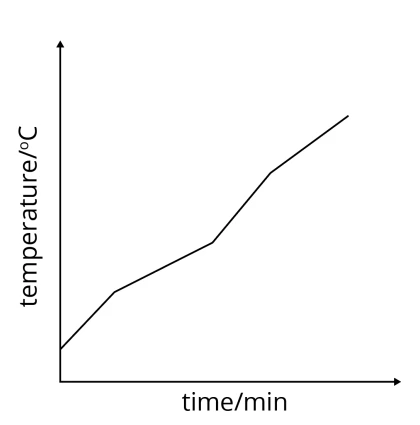
In the heating curve above, a substance is heated until it undergoes both melting and boiling. This substance melts and boils at fixed temperatures, as shown by the two constant temperature lines (horizontal lines).
The boiling point of pure water is 100ºC, while the melting point of ice is 0ºC.
-
Impure substance
- Melting point: An impure substance will melt over a range of temperatures.
- Boiling point: An impure substance will boil over a range of temperatures.
As seen from the heating curve below, when the impure substance is heated until it melts, the melting process does not occur at a fixed temperature. Similarly, when heating of the impure substance continues, the boiling process also does not occur at a fixed temperature.

Effects of impurities on melting and boiling points
We have seen earlier that the presence of impurities in a substance will affect its melting and boiling points.
- Impurities lower the melting point of a solid. Impurities also cause melting to occur over a range of temperatures.
- Impurities increase the boiling point of a liquid. Impurities also cause boiling to occur over a range of temperatures.
For example, when a small amount of salt is added to pure water, this mixture will now boil over a range of temperatures above 100ºC.
On the other hand, when a small amount of salt is added to ice, it will now melt over a range of temperatures below 0ºC.
What is a Mixture?
A mixture is made up of two or more substances that are not chemically combined together.
|
Image Credit: unsplash - Aliet Kitchen |
Image Credi: unsplash - Richard Bell |
Examples:
- Air is a mixture because it is made up of different types of gases, mainly nitrogen, oxygen, carbon dioxide, argon, as well as water vapour. These substances are not chemically combined together.
- Orange juice is a mixture because it consists of particles of solid (pulp) with liquid (juice). When you filter it, you can separate the pulp from the juice itself.
- Muddy water is also a mixture because it contains water, soil, mud, and many other components present within it, which are not chemically combined with one another.
How do we separate mixtures into their components?
Physical separation techniques are used to separate mixtures.
Examples of physical separation techniques:
- filtration
- evaporation to dryness
- crystallisation
- sublimation
- using a magnet
- simple distillation
- fractional distillation
- separating funnel
- chromatography
Methods of separating and purifying mixtures of solid and liquid
Filtration
Filtration is used to separate insoluble solid particles from a liquid.
A mixture of sand and water can be separated by filtration.
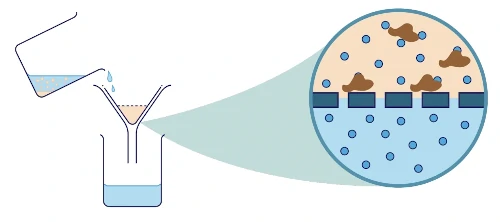
A piece of filter paper is used in this separation method. It is lined on the inner side of the filter funnel.
A filter paper has tiny pores that only allow very small tiny substances (in this case, water molecules) to pass through.
However, the insoluble particles like sand are too large to pass through the pores of the filter paper, hence, they will be left behind. The insoluble solid substance left behind on the filter paper is called the residue.
On the other hand, the soluble substance that passes through the filter paper and collects in a conical glass or a beaker is known as the filtrate.
Try It Yourself
The table shows some information about the solubilities of three solids.
| Solid | Soluble In Water | Soluble In Ethanol |
|---|---|---|
| P | No | Yes |
| Q | Yes | No |
| R | No | No |
Can a mixture containing P and Q be separated by filtration when water is added?
Answer:
Yes. Since solid P is insoluble in water while solid Q is soluble in the same solvent, filtration can be performed to separate the two solids.
P will be the residue left behind in the filter paper, while Q will be the filtrate.
Can a mixture containing P and R be separated by filtration when ethanol is added?
Answer:
Yes. Since solid P is soluble in ethanol while solid R is insoluble in the same solvent, filtration can be performed to separate the two solids.
R will be the residue left behind in the filter paper, while P will be the filtrate.
Evaporation to Dryness
Evaporation to dryness is used to obtain a soluble solid from a solution by heating the solution until all the water has boiled off.
The solid has to be heat-stable, so that the solid remains as crystalline residue after the solvent has evaporated.
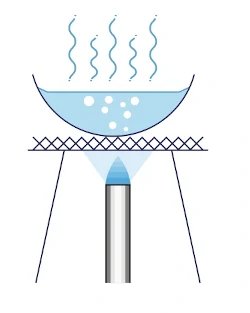
One common example of a mixture separated by evaporation to dryness is seawater. To obtain the sodium chloride salt (soluble solid) from the seawater, we can carry out evaporation. As sodium chloride is heat-stable, when all the water has been evaporated off, all that will be left on the evaporating dish are the salt crystals.
Note:
Not all soluble substances can be obtained by evaporation to dryness. If the solid substance is not heat-stable, strong heating to dryness will decompose it.
For example, if you want to obtain sugar from a sugar solution, strong heating of the sugar solution to dryness will cause the sugar inside your sugar solution to decompose to carbon. Hence, crystallisation should be used instead.
Crystallisation
Crystallisation is used for obtaining a soluble solid from its solution, where the solid is not heat-stable.

One common example is to obtain copper(II) sulfate crystals from its solution.
Steps followed in crystallisation
- The first step in crystallisation is to heat the impure solution until it becomes saturated (or two-thirds of its solvent has evaporated).
- Once we obtain the saturated solution, we allow it to cool. As the saturated solution cools down, you can observe the formation of crystals at the bottom of the evaporating dish. The size of the crystals formed depends on how the cooling is carried out. When the cooling takes place slowly, the crystals formed will be large.
- Filter to remove any remaining solution and to collect the crystals. Wash the crystals with a small amount of cold distilled water. This is to remove any other impurities present.
- The final step is to dry the crystals between sheets of filter paper.
Question 1:
Which of the following substances is a pure substance and not a mixture?
- common salt
- milk
- petrol
- soap
Answer:
A) common salt
Explanation:
Common salt, also known as sodium chloride, is a compound consisting of sodium and chlorine atoms chemically bonded together.
Question 2:
Which of the following properties indicates that a solid is pure?
- It burns completely in oxygen leaving no residue.
- It has a fixed melting point.
- It has a fixed volume.
- It melts over a range of temperatures.
Answer:
B) It has a fixed melting point.
Explanation:
Pure substances melt and boil at a fixed temperature, while impure substances melt and boil over a range of temperatures.
Question 3:
The boiling point of substance Y is 980ºC and is soluble in water. Which of the following is most likely the boiling point of water when a small quantity of Y is added to it?
- 97ºC
- 103ºC
- 978ºC
- 982ºC
Answer:
B) 103ºC
Explanation:
The boiling point of water is 100ºC.
When a small amount of impurity (substance Y) is added to pure water, the boiling point of water increases above 100ºC.
Conclusion
In this article, we have looked at the differences between pure and impure substances, as well as the methods to test the purity of substances.
Physical separation techniques can be used to separate a mixture to obtain the pure substances again.
We gained insight into some of the separation techniques used to separate a mixture of solid and liquid, such as filtration, evaporation to dryness and crystallisation.


 SG
SG  VN
VN