GCE O-Level 2025 Syllabus
GCE 2025 O-level syllabus overview
When preparing for an important paper such as the O Level examination, it is important to understand not only the topics in the subject but also the details of the paper. Over here, we have compiled a detailed list of what a student can expect to be tested on for O Level subjects. Remember, it is never too late to start preparing! All information has been from the SEAB website.
Learn more about each O Level subject:
English
In this English Language examination, students will be assessed on their ability to:
- Listen to, read, and view a wide array of literary and informational texts critically and accurately, demonstrating an understanding and appreciation of the text.
- Analyse different types of written and multimodal texts at basic, interpretive, and evaluative levels, including understanding how language creates an impact.
- Find main ideas and details in various texts, including those with pictures or videos. Also, demonstrate the ability to compile and summarise information from different sources.
- Listen to different audio texts and demonstrate your understanding in three ways:
- Be able to speak your thoughts and opinions clearly and smoothly to capture the listener's attention.
- Take part in a conversation and express your thoughts and opinions clearly.
Maths
The O Level Mathematics syllabus is intended to provide students with fundamental mathematical knowledge and skills. The content is organised into three strands:
- Number and Algebra
- Geometry and Measurement
- Statistics and Probability
When preparing for a critical examination, students must understand and revise all the topics in a subject. Over here, we have compiled a list of all the topics tested in the subject and what to expect in the papers.
Section 1: Numbers and algebra
Numbers and their operations
- Understand prime numbers and prime factorisation.
- Find the highest common factor (HCF) and lowest common multiple (LCM), squares, cubes, square roots, and cube roots using prime factorisation.
- Understand negative numbers, integers, rational numbers, real numbers, and their four operations.
- Perform calculations with a calculator.
- Represent and order numbers on the number line.
- Use symbols <, >, ⩽, ⩾ to compare numbers.
- Approximate and estimate numbers by rounding off to a required number of decimal places or significant figures and estimate results of computation.
- Use standard form A × \(10^n\), where n is an integer, and 1 ⩽ A < 10.
- Understand positive, negative, zero, and fractional indices.
- Apply laws of indices to solve mathematical problems.
Ratio and proportion
- Understand and apply ratios involving rational numbers.
- Simplify ratios to their simplest form.
- Understand map scales for both distance and area.
- Understand and solve problems using direct and inverse proportions.
Percentage
- Convert one quantity to a percentage of another quantity.
- Compare two quantities by percentage.
- Understand percentages greater than 100%.
- Calculate an increase or decrease in quantity by a given percentage.
- Solve problems involving reverse percentages.
Rate and speed
- Understand and apply the concepts of average rate, speed, and constant speed to problem-solving.
- Convert units of measurement, such as km/h to m/s, using conversion factors.
Algebraic expressions and formulae
- Use letters to represent numbers and interpret notations in algebraic expressions.
- Evaluate algebraic expressions and formulae in given situations.
- Translate simple real-world situations into algebraic expressions.
- Recognise and represent patterns/relationships by finding an algebraic expression for the nth term.
- Add and subtract linear expressions and simplify linear expressions.
- Use brackets and common factors.
- Know how to factorise linear expressions of the form \(ax + bx + kay + kby\).
- Expand the product of algebraic expressions.
- Change the subject of a formula.
- Find the value of an unknown quantity in a given formula using appropriate techniques.
- Use formulas such as \((a+b)^2 = a^2+2ab+b^2, (a-b)^2 = a^2-2ab+b^2\) and \(a^2-b^2 = (a+b)(a-b) \) to solve problems.
- Factorise quadratic expressions such as \(ax^2+bx+c\) and perform multiplication and division of simple algebraic fractions.
- Add and subtract algebraic fractions with linear or quadratic denominators.
Functions and graphs
- Understand and apply Cartesian coordinates in two dimensions.
- Use a graph of ordered pairs to represent the relationship between two variables.
- Understand linear functions (\(y = ax+b\)) and quadratic functions (\(y = ax^2+bx+c\)).
- Graph linear functions and understand the gradient of a linear graph as the ratio of vertical change to horizontal change (positive and negative gradients).
- Graph quadratic functions and identify their properties, including positive or negative coefficient of \(x^2\), maximum and minimum points, and symmetry.
- Sketch graphs of quadratic functions given in the form
- \(y = (x - p)^2 + q\)
- \( y = - (x - p)^2 + q\)
- \(y = (x - a)(x - b)\)
- \(y = - (x - a)(x - b)\).
- Graph power functions of the form \(y= ax^n\), where \(n = -2, -1, 0, 1, 2, 3,\) and simple sums of not more than three of these.
- Graph exponential functions \( y=ka^x\), where a is a positive integer.
- Estimate the gradient of a curve by drawing a tangent.
Equations and inequalities
- Solve linear equations with one variable.
- Solve simple fractional equations that can be reduced to linear equations.
- Solve simultaneous linear equations in two variables by substitution and elimination methods and graphical methods.
- Solve quadratic equations in one unknown by factorisation, use of formula, completing the square for \(y = x^2+px+q\), and graphical method.
- Solve fractional equations that can be reduced to quadratic equations.
- Formulate equations to solve problems.
- Solve linear inequalities in one variable, and represent the solution on the number line.
Set language and notation
- Usage of set language.
- Union and intersection of 2 sets.
- Venn diagrams.
Matrices
- Display of information in the form of a matrix of any order.
- Interpret the data in a given matrix.
- Calculate the product of a scalar quantity and a matrix.
- Solve problems involving the calculation of the sum and product of two matrices.
Section 2: Geometry and measurement
Angles, Triangles, and Polygons
- Identify and understand right, acute, obtuse, and reflex angles.
- Recognise vertically opposite angles, angles on a straight line, and angles at a point.
- Understand angles formed by two parallel lines and a transversal: corresponding angles, alternate angles, and interior angles.
- Explore properties of triangles, special quadrilaterals, and regular polygons (pentagon, hexagon, octagon, and decagon), including symmetry properties.
- Categorise special quadrilaterals based on their properties.
- Calculate the angle total of the interior and exterior angles of any convex polygon.
- Construct simple geometrical figures from given data, including perpendicular bisectors and angle bisectors, using compasses, rulers, set squares, and protractors where appropriate.
Congruence and similarity
- Identify congruent figures.
- Recognise similar figures.
- Explore properties of similar triangles and polygons.
- Understand the enlargement and reduction of a plane figure.
- Interpret scale drawings.
- Examine properties and construction of perpendicular bisectors of line segments and angle bisectors.
- Determine whether two triangles are congruent or similar.
- Calculate the ratio of areas of similar plane figures.
- Determine the ratio of volumes of similar solids.
- Solve simple problems involving similarity and congruence.
Properties of circles
- Symmetry properties of circles:
- Equal chords are equidistant from the centre.
- The perpendicular bisector of a chord passes through the centre.
- Tangents from an external point are of equal length.
- The line joining an external point to the centre of the circle bisects the angle between the tangents.
- Angle properties of circles:
- An angle in a semicircle is a right angle.
- The angle between a tangent and the radius of a circle is a right angle.
- The angle at the centre is twice the angle at the circumference.
- Angles in the same segment are of equal measure.
- Angles in opposite segments are supplementary.
Pythagoras’ theorem and trigonometry
- Use of Pythagoras’ theorem.
- Determining whether a triangle is right-angled given the lengths of three sides.
- Use of trigonometric ratios (sine, cosine and tangent) of acute angles to calculate unknown sides and angles in right-angled triangles.
- Extending sine and cosine to obtuse angles.
- Use of the formula \(\begin{align}{1\over2}ab\sin C\end{align}\) for the area of a triangle.
- Use of sine rule and cosine rule for any triangle.
- Solve problems in two and three dimensions, including those involving angles of elevation and depression and bearings.
Mensuration
- Calculate the area of parallelogram and trapezium.
- Solve problems related to the perimeter and area of combined plane figures.
- Determine the volume and surface area of a cube, cuboid, prism, cylinder, pyramid, cone, and sphere.
- Convert \(cm^2\) and \(m^2\), as well as \(cm^3\) and \(m^3.\)
- Solve problems related to the volume and surface area of combined solids.
- Determine the arc length, sector area, and area of a segment of a circle.
- Use radian measurement of angle, including conversion between radians and degrees.
Coordinate Geometry
- Determine the gradient of a straight line by using the coordinates of two points on a line.
- Ascertain the length of a line segment using the coordinates of its endpoints.
- Interpret and determine the equation of a straight-line graph in the format of y=mx+c.
- Solve geometrical problems that involve the application of coordinates.
Vectors in two dimensions
- Use of vector notations.
- Represent a vector as a directed line segment.
- Perform translation by a vector.
- Understand position vectors.
- Calculate the magnitude of a vector.
- Use of sum and difference of two vectors to express given vectors in terms of two coplanar vectors.
- Multiply of a vector by a scalar.
- Solve geometric problems involving the use of vectors.
Section 3: Statistics and probability
Data handling and analysis
- Grasp simple concepts in collecting, classifying, and tabulating data.
- Analyse and interpret various forms of statistical representations:
- Tables
- Bar graphs
- Pictograms
- Line graphs
- Pie charts
- Dot diagrams
- Histograms with equal class intervals
- Stem-and-leaf diagrams
- Cumulative frequency diagrams
- Box-and-whisker plots.
- Understand the purposes, uses, advantages, and disadvantages of different forms of statistical representations.
- Draw simple inferences from statistical diagrams.
- Explain why a given statistical diagram may lead to the misinterpretation of data.
- Understand mean, mode, and median as measures of central tendency for a set of data.
- Recognise the purposes and use of mean, mode, and median.
- Calculate the mean for grouped data.
- Understand quartiles and percentiles.
- Explore range, interquartile range, and standard deviation as measures of spread for a set of data.
- Calculate the standard deviation for a set of data (grouped and ungrouped).
- Use the mean and standard deviation to compare two sets of data.
Probability
- Demonstrate an understanding of probability as a measure of likelihood.
- Calculate the probability of individual events (including listing all possible outcomes in a simple probability scenario to determine likelihood).
- Determine the probability of combined events (including using possibility diagrams and tree diagrams when applicable).
- Apply addition and multiplication of probabilities (for events that are mutually exclusive or independent).
A. Maths
The O-level Additional Mathematics syllabus is designed to equip students with fundamental mathematical knowledge for A-level H2 Mathematics. The content is organised into three strands:
- Algebra,
- Geometry and Trigonometry, and,
- Calculus.
Section 1: Algebra
Quadratic functions
- Find the maximum or minimum value of a quadratic function using the method of completing a square.
- Understand the conditions for \(y=ax^2+bx+c\) to be always positive (or always negative).
- Utilise quadratic functions as models.
Equations and inequalities
- Conditions for a quadratic equation to have:
- (i) two real roots, (ii) two equal roots, (iii) no real roots.
- Determine conditions for a given line to:
- (i) intersect a given curve, (ii) be tangent to a given curve, (iii) not intersect a given curve.
- Solve simultaneous equations in two variables by substitution, with one of the equations being a linear equation.
- Solve quadratic inequalities, and represent the solution on the number line.
Surds
- Perform the four operations on surds, including rationalising the denominator.
- Solving equations involving surds.
Polynomials and partial fractions
- Perform multiplication and division of polynomials.
- Apply the remainder and factor theorems, including factorising polynomials and solving cubic equations.
- Utilise the following polynomial factorisations:
- \(a^3+b^3=(a+b)(a^2-ab+b^2)\)
- \(a^3-b^3=(a-b)(a^2+ab+b^2)\).
- Work with partial fractions with cases where the denominator is not more complicated than:
- \((ax+b)(cx+d)\)
- \((ax+b)(cx+d)^2\)
- \((ax+b)(x^2+c^2)\).
Binomial expansions
- Use of the Binomial Theorem for positive integer n.
- Use of the notations \(\displaystyle n!\) and \(\displaystyle \binom{n}{r}\).
- Use of the general term \(\displaystyle \binom{n}{r}a^{n-r}b^r, 0\le r \le n\) (knowledge of the greatest term and properties of the coefficients is not required).
Exponential and logarithmic functions
- Understand exponential and logarithmic functions \(a^x\) , \(e^x\) , \(log_{a}x\), \(\ln x\) and their graphs, including:
- laws of logarithms
- equivalence of \(y=a^x\)and \(x=\log_{a}y\)
- change of base of logarithms.
- Simplify expressions and solve simple equations involving exponential and logarithmic functions.
- Use exponential and logarithmic functions as models.
Section 2: Geometry and trigonometry
Trigonometric functions, identities, and equations
- Understand six trigonometric functions for angles of any magnitude (in degrees or radians).
- Determine principal values of \(\displaystyle \sin^{-1}x, \cos^{-1}x, \tan^{-1}x\).
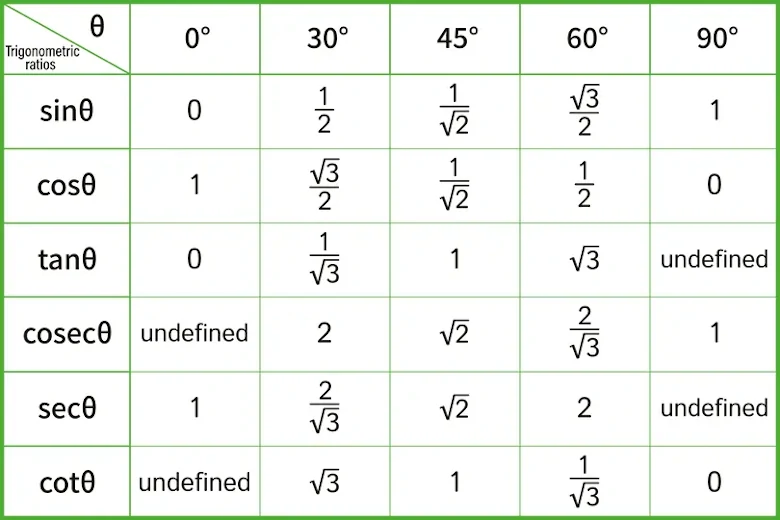
- Identify exact values of the trigonometric functions for special angles \((30^\circ, 45^\circ, 60^\circ)\) or \(\begin{align}\bigg(\frac{\pi}{6},\frac{\pi}{4},\frac{\pi}{3}\bigg)\end{align}\).
- Explore amplitude, periodicity and symmetries related to sine and cosine functions.
- Graphs of \(\displaystyle y=a \sin(bx)+c, y=a \sin(\frac{x}{b})+c, y=a \cos(bx)+c, y=a \cos(\frac{x}{b})+c, y=a \tan(bx)\) where \(\displaystyle {a}\) is real, \(\displaystyle {b}\) is a positive integer and \(\displaystyle {c}\) is an integer.
- Utilise the following trigonometric relationships:
\(\begin{align*} &\frac{\sin A}{\cos A} = \tan A, \frac{\cos A}{\sin A} = \cot A, \sin^2A+cos^2A=1 \\ \\ &\sec^2A=1+\tan^2A, \DeclareMathOperator\cosec{cosec}\cosec^2A=1+\cot^2A \end{align*}\) - Apply the expressions of \(\displaystyle \sin(A\pm B), \cos(A\pm B), \tan(A \pm B)\).
- Use the formulae for \(\displaystyle \sin2A, \cos2A, \tan2A\).
- Express \(\displaystyle a\cos\theta+b\sin\theta\) in the form \(\displaystyle R\cos(\theta\pm\alpha) \text{ or } R\sin(\theta\pm\alpha)\).
- Simplify trigonometric expressions.
- Solve simple trigonometric equations in a given interval (excluding general solution).
- Prove simple trigonometric identities.
- Use trigonometric functions as models.
Coordinate geometry in two dimensions
- Condition for two lines to be parallel or perpendicular.
- Midpoint of a line segment.
- Area of a rectilinear figure.
- Coordinate geometry of circles in the form:
\((x-a)^2+(y-b)^2=r^2\)
\(x^2+y^2+2gx+2fy+c=0\)
(excluding problems involving two circles) - Transformation of given relationships, including \(y=ax^n\) and \(y=kb^x\), to linear form to determine the unknown constants from a straight-line graph.
Proofs in plane geometry
- Understand the properties of parallel lines cut by transversal, perpendicular lines, and angle bisectors.
- Apply geometric principles to triangles, special quadrilaterals, and circles.
- Recognise congruent and similar triangles.
- Utilise the Midpoint Theorem.
- Apply the Tangent-Chord Theorem (Alternate Segment Theorem).
Section 3: Calculus
Differentiation and integration
- Understand the derivative of \(f(x)\) as the gradient of the tangent to the graph of \(y=f(x)\) at a point.
- Recognise the derivative as the rate of change.
- Use of standard notations
- \(\begin{align}f'(x), f''(x), \frac{dy}{dx}, \frac{d^2y}{dx^2}\bigg[=\frac{d}{dx}(\frac{dy}{dx})\bigg]\end{align}\).
- Find derivatives of \(x^n\) , for any rational \(n\), \(\sin x, \cos x, \tan x, e^x, \ln x\), together with constant multiples, sums. and differences.
- Calculate the derivatives of products and quotients of functions
- Apply the Chain Rule.
- Understand increasing and decreasing functions.
- Identify stationary points (maximum and minimum turning points and stationary points of inflexion).
- Use of the second derivative test to discriminate between maxima and minima.
- Apply differentiation to gradients, tangents and normals, connected rates of change and maxima and minima problems.
- Recognise integration as the reverse of differentiation.
- Integrate \(X^n\)for any rational \(\displaystyle n, \sin x, \cos x, \sec^2x, e^x\) together with constant multiples, sums and differences.
- Integrate \((ax+b)^n\) + for any rational \(n\), \(\sin(ax+b), \cos(ax+b)\) and \(e^{ax+b}\).
- Understand the definite integral as an area under a curve.
- Evaluate definite integrals.
- Find the area of a region bounded by a curve and line(s) (excluding the area of the region between 2 curves).
- Find areas of regions below the x-axis.
- Apply differentiation and integration to problems involving displacement, velocity, and acceleration of a particle moving in a straight line.
Physics
Section 1: Measurement
- Physical quantities, units, and measurement
- Physical quantities and SI units
- Measurement
- Scalars and vectors
Section 2: Newtonian Mechanics
- Kinematics
- Speed, velocity, and acceleration
- Graphical analysis of motion
- Free-fall
- Dynamics
- Types of forces
- Mass, weight, and gravitational field
- Newton's laws of motion
- Effects of resistive forces of motion
- Turning effects of forces
- Moments
- Equilibrium
- Centre of gravity and stability
- Pressure
- Pressure
- Density and fluid pressure
- Energy
- Energy stores and transfers
- Work, power, and efficiency
- Energy resources
Section 3: Thermal Physics
- Kinetic particle model of matter
- States of matter
- Kinetic particle model
- Thermal processes
- Thermal equilibrium
- Conduction
- Convection
- Radiation
- Thermal properties of matter
- Internal energy
- Specific heat capacity
- Melting, boiling, and evaporation
- Specific latent heat
Section 4: Waves
- General properties of waves
- Describing wave motion
- Wave properties
- Sound
- Electromagnetic spectrum
- Properties of electromagnetic waves
- Applications of electromagnetic waves
- Effects of electromagnetic waves on cells and tissues
- Light
- Reflection of light
- Refraction of light
- Thin converging lenses
Section 5: Electricity and magnetism
- Static electricity
- Electric charge
- Electric field
- Dangers and applications of electrostatic charging
- Current of electricity
- Conventional current and electron flow
- Electromotive force and potential difference
- Resistance
- D.C. circuits
- Circuit diagrams
- Series and parallel circuits
- Action and use of circuit components
- Practical electricity
- Electrical working, power, and energy
- Dangers of electricity
- Safe use of electricity at home
- Magnetism
- Laws of magnetism
- Magnetic properties of matter
- Magnetic field
- Electromagnetism
- Magnetic effect of a current
- Force on a current-carrying conductor
- The d.c. motor
- Electromagnetic induction
- Principles of electromagnetic induction
- The a.c. generator
- The transformer
Section 6: Radioactivity
Chemistry
Section 1: Matter - Structures and Properties
- Experimental chemistry
- Experimental design
- Methods of purification and analysis
- The particulate nature of matter
- Kinetic particle theory
- Atomic structure
- Chemical bonding and structure
- Ionic bonding
- Covalent bonding
- Metallic bonding
- Structure and properties of materials
Section 2: Chemical reactions
- Chemical calculations
- Formulae and equation writing
- The mole concept and stoichiometry
- Acid-base chemistry
- Acids and bases
- Salts
- Ammonia
- Qualitative analysis
- Redox chemistry
- Oxidation and reduction
- Electrochemistry
- Patterns in the periodic table
- Periodic table trends
- Group properties
- Transition elements
- Reactivity series
- Chemical energetics
- Rate of reactions
Section 3: Chemistry in a sustainable world
- Organic chemistry
- Maintaining air quality
Biology
Section 1: Cells and the chemistry of life
- Cell structure and organisation
- Plant and animal cells
- Cell specialisation
- Movement of substances
- Diffusion
- Osmosis
- Active transport
- Biological molecules
- Carbohydrates, fats, and proteins
- Enzymes
- Nutrition in humans
- Human digestive system
- Physical and chemical digestion
- Absorption and assimilation
- Transport in humans
- Parts and functions of the circulatory system
- Blood
- Heart and cardiac cycle
- Coronary heart disease
- Respiration in humans
- Human gas exchange
- Cellular respiration
- Excretion in Humans
- Structure and function of kidneys
- Kidney dialysis
- Homeostasis, co-ordination, and response in humans
- Principles of homeostasis
- Hormonal control
- Nervous control
- Infectious diseases in humans
- Organisms affecting human health
- Influenza and pneumococcal disease
- Prevention and treatment of infectious diseases
- Nutrition and transport in flowering plants
- Plant structure
- Photosynthesis
- Transpiration
- Translocation
- Organisms and their environment
- Energy flow
- Food chains and food webs
- Carbon cycle and global warming
- Effects of man on the ecosystem
- Conservation
- Molecular genetics
- The structure of DNA
- From DNA to proteins
- Genetic engineering
- Reproduction
- Asexual reproduction
- Cell division
- Sexual reproduction in flowering plants
- Sexual reproduction in humans
- Sexually transmitted diseases
- Inheritance
- The passage of genetic information from parent to offspring
- Monohybrid crosses
- Variation
- Natural selection


 SG
SG  VN
VN