The Mole
In this article we are going to learn about the concept of the Mole. We will be covering the following learning outcomes:
- Define relative atomic mass
- Define relative molecular mass & relative formula mass
- Calculate relative molecular mass or relative formula mass of a substance
- Define the term mole
- Convert number of particles into number of moles of particles and vice versa
This article is written to meet the requirements of the Upper Secondary Chemistry syllabus in Singapore.
Relative Atomic Mass
By definition, the relative atomic mass of an atom (Ar) is the average mass of one atom of that element compared to \(\displaystyle{1\over 12}\) of the mass of one carbon-12 atom.
Question:
Why do we use the mass of a carbon-12 and not the actual mass of an atom of that element?
Answer:
To answer this, let us take a look at the next section.
Atomic Mass Unit
All the particles in an atom are very light, so light that it is not convenient and practical to express their masses in grams or kilograms in calculations.
It is easier to measure their masses against a standard unit, called the atomic mass unit (amu).
\(\small\displaystyle{\textsf{1 amu} = \mathsf{1.67 \times 10^{-27} kg}}\)
1 amu is \(1\over 12\) the mass of a carbon-12 atom. The mass of this carbon was determined accurately by experiment using a modern instrument called mass spectrometer.
\(\begin{align} \small \displaystyle{\textsf{Relative Aatomic Mass} = \frac{\textsf{Average mass of an atom of element}}{{\frac {1}{12}} \times \textsf{Mass of an atom of carbon-12}}} \end{align}\)
For example, one atom of oxygen was found to be 16 times heavier than \(\displaystyle{1\over 12}\) of an atom of carbon-12. Thus, oxygen has a relative atomic mass of 16.
As the relative atomic mass is a ratio, it does not have a unit.
Why is the Ar of Chlorine a decimal?
Some elements, such as chlorine, occur as mixtures of isotopes. For these elements, their atoms will have different relative masses.
For ease of calculation, the relative atomic mass will be calculated based on the relative percentage abundance of the isotopes.
Recall that isotopes are atoms of the same element with the same number of protons and electrons, but different numbers of neutrons.
There are two different isotopes of chlorine: 35Cl and 37Cl.
Since we cannot account for both of these isotopes individually, hence we take the average of both of these atomic masses.
Worked example:
Chlorine has two isotopes, 35Cl and 37Cl, with respective abundance of 75% and 25%. Determine the relative atomic mass of chlorine.
Solution:
Relative atomic mass of chlorine
\(\displaystyle{=}\) Sum of [relative abundance × isotopic mass]
\(\displaystyle{=\frac {75}{100} \times 35 + \frac {25}{100} \times 37}\)
\(\displaystyle{= 35.5}\)
Question 1:
Chemists discovered that the neon element has three isotopes. They are neon-20, neon-21 and neon-22, with respective abundances of 90.92%, 0.26% and 8.82%.
Determine the Ar of neon. (* Give your answer correct to 3 s.f.)
- 21.1
- 20.2
- 20.9
- 21.9
Solution:
(B) 20.2
Explanation:
Ar = Sum of [relative abundance \(\times\) isotopic mass]
\(\displaystyle{= \frac {90.92}{100} \times 20 + \frac{0.26}{100} \times 21 + \frac{8.82}{100} \times 22}\)
\(= 20.179\)
\(= 20.2 \; \text{(to 3.s.f.)}\)
Question 2:
The diagram shows the mass spectrum of an unknown sample. Use the data to calculate the relative atomic mass of the sample.
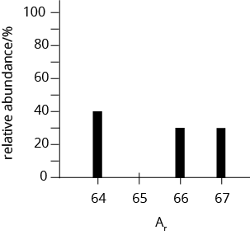
- 65.00
- 65.25
- 65.50
- 65.66
Solution:
(C) 65.50
Explanation:
Ar = Sum of [relative abundance \(\times\) isotopic mass]
= \(\frac {40} {100}\; ×\; 64 \;+\; \frac {30} {100}\; ×\; 66 \;+\; \frac {30} {100}\; ×\; 67\)
= 65.50
Question 3:
Potassium has 2 major isotopes. They are 39K and 41K. If the relative atomic mass of naturally occurring potassium is 39.14, what are the relative abundances of 39K and 41K?
- 7% of 39K & 93% of 41K
- 25% of 39K & 75% of 41K
- 75% of 39K & 25% of 41K
- 93% of 39K & 7% of 41K
Solution:
(D) 93% of 39K & 7% of 41K
Explanation:
We can perform a calculation for each of the options. So when the relative abundance of 39K and 41K are 93% and 7% respectively, the relative atomic mass of potassium is:
Ar = Sum of [relative abundance \(\times\) isotopic mass]
= \(\frac {93} {100}\; ×\; 39 \;+\; \frac {7} {100}\; ×\; 41 \)
= 39.14
Relative Molecular Mass
By definition, the relative molecular mass of a molecular substance (Mr) is the average mass of one molecule of that element or compound compared to \(1\over 12\) of the mass of one carbon-12 atom.
To compute the relative molecular mass of a substance, we can simply add the relative atomic masses of all the atoms in its chemical formula.
Worked example:
Calculate the relative molecular mass of sulfuric acid, H2SO4
Solution:
Relative molecular mass of sulfuric acid, H2SO4
= (2 × Ar of H atom ) + (1 × Ar of S atom) + (4 × Ar of O atom)
= (2 × 1 ) + (1 × 32) + (4 × 16)
= 98
Relative formula Mass
By definition, the relative formula mass of an ionic compound (Mr) is the average mass of one unit of that ionic compound compared to \(1\over 12\) of the mass of one carbon-12 atom.
To compute the relative formula mass of a substance, we can simply add the relative atomic masses of all the atoms in its chemical formula.
Worked example:
Calculate the relative formula mass of magnesium chloride, MgCl2
Solution:
Relative formula mass of magnesium chloride, MgCl2
= (1 × Ar of Mg atom ) + (2 × Ar of Cl atom)
= (1 × 24) + (2 × 35.5)
= 95
Question 4:
Calculate the relative molecular mass of C3H7COOH.
- 72
- 76
- 87
- 88
Solution:
(D) 88
Explanation:
Relative molecular mass of C3H7COOH
= (4 × Ar of C atom ) + (8 × Ar of H atom) + (2 × Ar of O atom)
= (4 × 12) + (8 × 1) + (2 × 16)
= 88
Question 5:
Calculate the relative formula mass of Fe(NO3)3
- 166
- 194
- 214
- 242
Solution:
(D) 242
Explanation:
Looking at the chemical formula of iron(III) nitrate, Fe(NO3)3, there are 1 Fe atom, 3 N atoms, and 9 O atoms.
Relative formula mass of Fe(NO3)3
= (1 × Ar of Fe atom ) + (3 × Ar of N atom) + (9 × Ar of O atom)
=(1 × 56 ) + (3 × 14) + (9 × 16)
= 242
Question 6:
Calculate the relative formula mass of CuSO4.7H2O.
- 190
- 286
- 2256
- 20160
Solution:
(B) 286
Explanation:
Looking at the chemical formula of hydrated copper(II) sulfate, CuSO4.7H2O, there are 1 Cu atom, 1 S atom, 4 O atoms, and 7 molecules of H2O.
Relative formula mass of CuSO4.7H2O
= (1 × Ar of Cu atom ) + (1 × Ar of S atom) + (4 × Ar of O atom) + (7 × Mr of H2O)
=(1 × 64) + (1 × 32) + (4 × 16) + [7 × (2 × 1 + 16)]
= 286
Mole
Imagine you are at the supermarket. Many items can be quantified by a unit.
For example:
1 kg of rice
1 litre of oil
1 pair of socks
1 dozen of eggs
However, how do you quantify particles such as atoms which are so small and numerous?
We will use the unit called mole.
One mole of a substance contains the same number of particles as the number of atoms in 12 g of carbon-12.
\(\displaystyle{\textsf{One mole of particles} = 6 × 10^{23} \textsf{particles}}\)
This value is also called Avogadro's constant or Avogadro’s number.
For Example:
1 mole of Cu atoms = 6 × 1023 Cu atoms
1 mole of H2O molecules = 6 × 1023 H2O molecules
Converting between the number of moles and number of particles, we can apply the following formula:
\(\displaystyle{\textsf{Number Of Moles}} = {\frac{\textsf{Number Of Particles}} {6 \times 10^{23}}}\)
Worked Example:
One molecule of H2O contains 2 H atoms and 1 O atom. Hence, in one mole of H2O molecules, determine the number of H atoms and O atoms.
Solution:
One mole of H2O molecules = 6 × 1023 H2O molecules
If each molecule of H2O contains 2 H atoms, then number of H atoms
\(= 6 × 10^{23}× 2\)
\(= 1.2 × 10^{24}\)
If each molecule of H2O contains 1 O atom, then number of O atoms
\(= 6 × 10^{23} × 1\)
\(= 6 × 10^{23}\)
Question 7:
Calculate the number of sulfur atoms in 0.25 mole of sulfur.
- 6 × 1023
- 3 × 1023
- 1.5 × 1023
- 0.25 × 1023
Solution:
(C) 1.5 × 1023
Explanation:
\(\text{Number of sulfur atoms }\)
\(= \text{( Number of moles of sulfur )} × 6 × 10^{23}\)
\(= 0.25 × 6 × 10^{23}\)
\(= 1.5 × 10^{23}\)
Question 8:
Calculate the number of chlorine atoms in 0.04 mole of chlorine gas.
- 0.24 × 1023
- 0.48 × 1023
- 2.4 × 1023
- 0.48 × 1024
Solution:
(B) 0.48 × 1023
Explanation:
\(\text{Number of chlorine gas molecules }\)
\(= \text{Number of moles of chlorine molecules} \times 6 \times 10^{23}\)
\(= 0.04 × 6 × 10^{23}\)
\(= 0.24 × 10^{23}\)
Each chlorine gas molecule contains 2 chlorine atoms.
\(\text{Number of chlorine atoms}\)
\(= 0.24 × 10^{23} \times 2\)
\(= 0.48 × 10^{23}\)
Conclusion
In this chapter, we have learned about the concepts of relative atomic mass, relative molecular mass as well as relative formula mass. We also learned how to calculate the relative molecular mass or relative formula mass by adding the relative atomic masses of all the atoms in the chemical formula of the substance. Lastly, we also learnt about mole as a unit of measurement and Avogadro’s constant, which allows us to convert between the number of moles and the number of particles.
Practice the multiple questions and examples given in the article to understand the concepts better. Keep Learning! Keep Improving!


 SG
SG  VN
VN