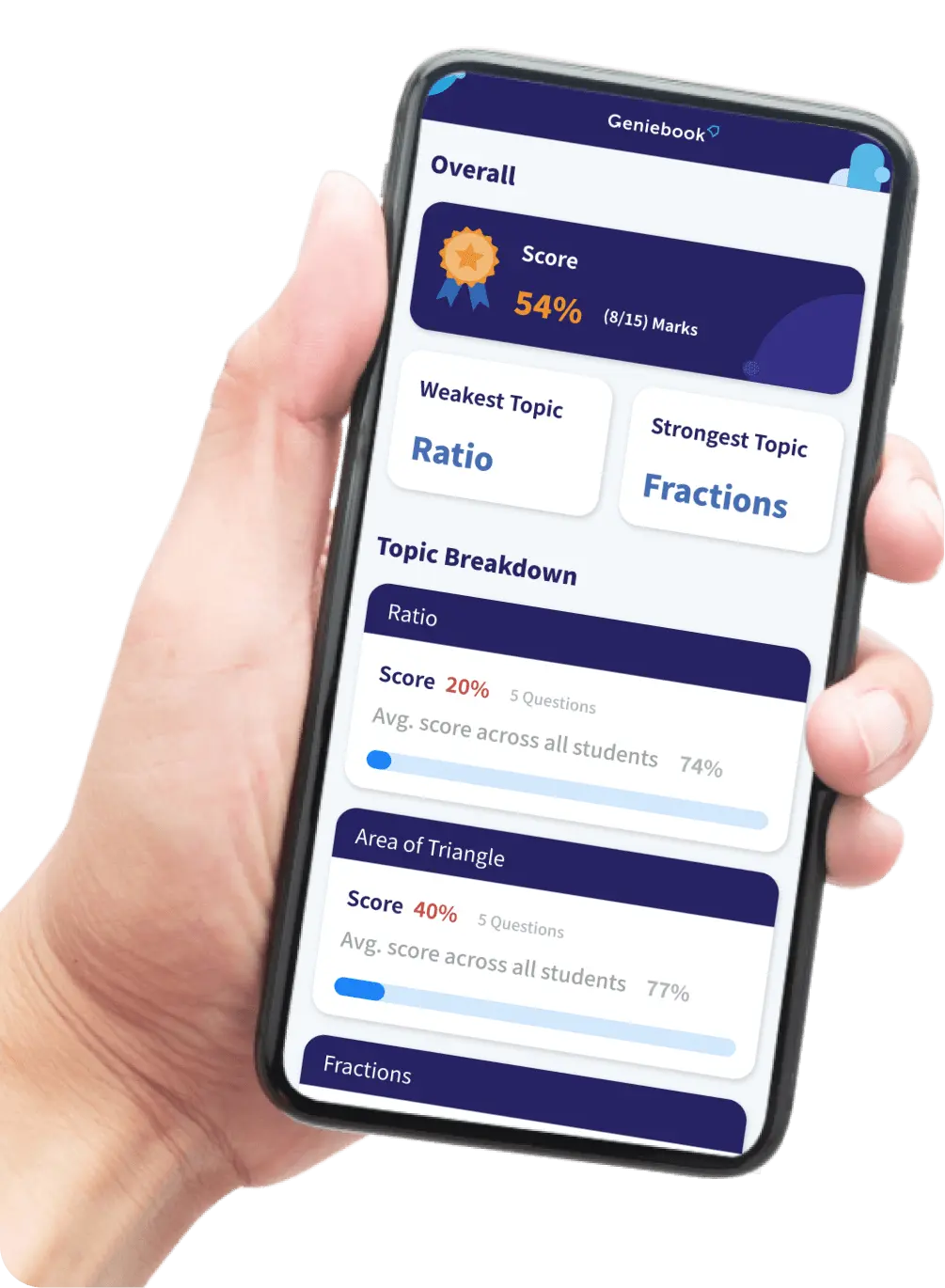
Temperature
In this article, we will be learning about temperature and the difference between temperature and heat. We will also be learning about how to calibrate a thermometer. This article has been written keeping in view the academic requirements of Secondary 3 Physics in Singapore.
What is Temperature?
Temperature is the measure of how hot or cold an object is.
The SI unit of temperature is Kelvin (K).
A commonly used unit of temperature is degree Celsius ( \(^{\circ}\)C ).
Temperature and Heat
|
Temperature |
Heat |
|---|---|
|
It refers to how hot or cold an object is |
It refers to the amount of thermal energy that is being transferred from a hotter to a colder region. |
|
The SI unit is Kelvin (K) |
The SI unit is Joule (J) |
Temperature and Heat
Indicate the net heat (energy) flow in the following diagrams:
Scenario 1:
The diagram above shows two objects, A and B. A is the hotter object and B is the colder object.
There is thermal energy transfer between the two objects A and B. However, the rate of thermal energy transfer from A to B is higher than that from B to A.
Since more energy is transferred from A to B per unit time, there is net heat flow from A to B.
Scenario 2:
Since both A and B are at the same temperature, there will be an equal rate of heat transfer between the two bodies. The net flow of heat between the two bodies will be zero.
Thermometric Substance
A thermometric substance is a substance that has a physical property that changes linearly as temperature changes.
Some examples of such physical property are
- Volume (of a fixed mass of liquid)
- Pressure (of gas thermometer)
- Resistance (of resistive wires)
- The electric potential difference between two metal junctions (of thermocouples)
Constructing a Temperature Scale
The steps to construct a temperature scale are as follows:
- Choose an appropriate thermometric substance.
- Calibrate the thermometer
- Choose two fixed points: lower fixed point (0\(^{\circ}\)C) and upper fixed point (100\(^{\circ}\)C) respectively.
- Set up the scale: divide the interval between the two fixed points into 100 equal parts. Each interval is 1\(^{\circ}\)C or 1 K.
Determining the Ice Point (Lower Fixed Point) using length \(\ell\) of mercury column
- Fully immerse the bulb of the thermometer into a funnel containing pure melting ice.
- When the mercury level in the thermometer stem is steady, make a mark \(\ell_{\circ}\) at that level on the stem.
This is called the ice point.
Determining the Steam Point (Upper Fixed Point) using length \(\ell\) of mercury column
- Insert the thermometer as shown. The bulb should be just above the boiling water.
- Make sure the thermometer is fixed in position throughout the experiment.
- When the mercury level in the thermometer stem is steady, make a mark \(\ell_{100}\) at that level on the stem.
This is the steam point.
Calculating temperature using length \(\ell\) of mercury column
Temperature is calculated using the equation below:
\(\begin{align} x = \frac {\ell_x -\ell_0}{\ell_{100}-\ell_0} \times 100^{\circ}C \end{align}\)
Where \(\ell_{0}\) and \(\ell_{100}\) are the ice and steam points respectively.
| \(\begin{align} \theta= \frac {\ell_{\theta} - \ell_0}{\ell_{100}-\ell_0} \times 100^{\circ}C \end{align}\) |
Practice Questions
Question 1:
The range of mercury in a glass thermometer is from 0\(^{\circ}\)C to 100\(^{\circ}\)C. The length of the mercury thread between the 0\(^{\circ}\)C and 100\(^{\circ}\)C marks on the stem is 20 cm. Determine the temperature when the length of the mercury thread is 12 cm from the 0\(^{\circ}\)C mark.
Solution:
By proportion,
\(\begin{align} 20 \text{ cm } ---------\;\; &100^{\circ}\text{C}\\[2ex] 12 \text{ cm } ---------\;\; &{\frac{12}{20} } \times 100^{\circ}C\\[2ex] &=60^{\circ}\text{C} \end{align} \)
Therefore,
\(\begin{align} \theta &= 60^{\circ}\text{C} + 0^{\circ}\text{C}\\[2ex] &= 60^{\circ}\text{C} \end{align}\)
Question 2:
The range of mercury in a glass thermometer is from −10\(^{\circ}\)C to 110\(^{\circ}\)C. The length of the mercury thread between the −10\(^{\circ}\)C to 110\(^{\circ}\)C marks on the stem is 20 cm. Determine the temperature when the length of the mercury thread is 12 cm from the −10\(^{\circ}\)C mark.
Solution:
By proportion,
\(\begin{align} 20 \text{ cm } ---------\;\; &120^{\circ}\text{C}\\[2ex] 12 \text{ cm } ---------\;\; &{\frac{12}{20} } \times 120^{\circ}C\\[2ex] &=72^{\circ}\text{C} \end{align} \)
Therefore,
\(\begin{align} \theta &= -10^{\circ}\text{C} + 72^{\circ}\text{C}\\[2ex] &= 62^{\circ}\text{C} \end{align}\)
Question 3:
The resistance of a piece of platinum wire is 450 Ω when placed in pure melting ice and 560 Ω when placed in steam above boiling water. Determine the temperature of a molten substance if the resistance of the wire is found to be 1260 Ω when placed in the substance.
Solution:
\(\begin{align} \theta &= \frac {R_{\theta} -R_0}{R_{100} - R_0} \times 100^{\circ}\text{C} \\[2ex] \theta &= \frac {1260 \,\Omega - 450\,\Omega}{560 \,\Omega - 450 \,\Omega} \times 100^{\circ}\text{C} \\[2ex] \theta &= \frac {810 \,\Omega}{110 \,\Omega} \times 100^{\circ}\text{C} \\[2ex] \theta &= 736^{\circ}\text{C} \\[2ex] \end{align} \)
Question 4:
The range of mercury in the glass thermometer is from −10\(^{\circ}\)C to 110\(^{\circ}\)C. The length of the mercury thread between −10\(^{\circ}\)C to 110\(^{\circ}\)C marks on the stem is 20 cm. Determine the temperature when the length of the mercury thread is 12 cm from the 110\(^{\circ}\)C mark.
Solution:
By proportion,
\(\begin{align} 20 \text{ cm } ---------\;\; &120^{\circ}\text{C}\\[2ex] \text{X cm } ---------\;\; &{\frac{20 -12}{20} } \times 120^{\circ}\text{C}\\[2ex] &=48^{\circ}\text{C} \end{align} \)
Therefore,
\(\begin{align} \theta &= -10^{\circ}\text{C} + 48^{\circ}\text{C}\\[2ex] &= 38^{\circ}\text{C} \end{align}\)
Conclusion
In this article we have learned about:
- Measurement of Temperature
- Calibrating a Thermometer
This article has been written keeping in mind the syllabus requirements of the Secondary 3 Physics lesson.
This article is very calculation intensive, so we would request you all to solve these questions repeatedly to get a better understanding.


 SG
SG  VN
VN