Periodic Table
In this article, we will be learning about the Periodic Table. These are the learning outcomes:
- Describe the Periodic Table as an arrangement of the elements according to proton number.
- Describe the change in metallic properties of the elements across a period of the Periodic Table.
- Describe the relationship between group number, number of valence electrons, and metallic/non-metallic properties.
- Describe the physical and chemical properties of Group I elements.
This article is specifically written to serve the requirements for the Upper Secondary Chemistry students in Singapore.
General features of Periodic Table
The Periodic Table is a list of elements arranged in order of increasing proton (atomic) numbers.
For example:
The first element in the Periodic Table is hydrogen (H) with a proton number of 1. The next element is helium (He) with a proton number of 2.
Advantages for arranging elements in the Periodic Table:
- It allows us to study and understand the relationship between various elements.
- It allows us to identify the properties of the elements easily.
- It allows us to predict the properties of an element.
The Periodic Table divides the elements into periods and groups.
A group is a vertical column of elements. It runs from top to bottom and there are eight groups of elements, numbered Ⅰ to Ⅶ, and 0.
A period is a horizontal row of elements. It runs from left to right and there are seven periods of elements, numbered 1 to 7.
The Periodic Table also classifies elements into metals and non-metals.
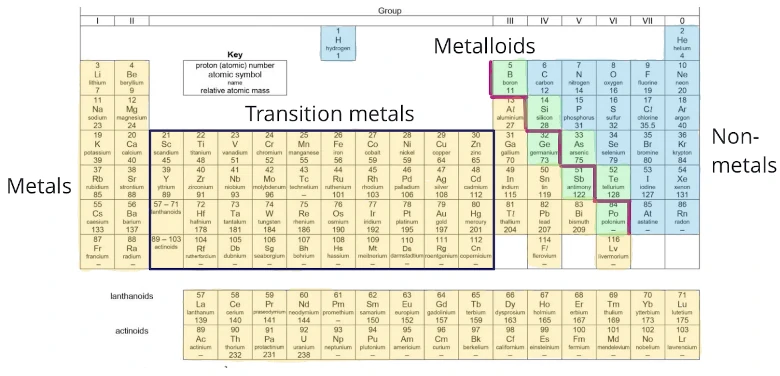
Image Credits: SEAB
A zig-zag line (red line) separates the metals (shaded in yellow) from the non-metals (shaded in blue) in the Periodic Table.
Metalloids are elements that have both metallic and non-metallic properties. They are elements located near the zig-zag line which separates the metals from the non-metals (shaded in green).
Periodic trends
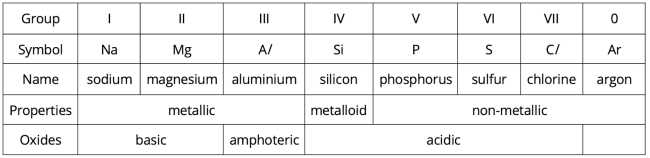
Let us examine the properties of elements across Period 3.
-
Metallic properties
Metals are found on the left-hand side of each period, while non-metals are found on the right-hand side.
From left to right, there is a decrease in metallic properties and an increase in non-metallic properties.
-
Nature of oxides
Metals, such as sodium and magnesium, form basic oxides. Some metals, such as aluminium, form amphoteric oxides. Non-metals, such as silicon, phosphorus, sulfur and chlorine, form acidic oxides.
Hence, we observe that as we go across the period, the nature of the oxides changes from basic to amphoteric to acidic.
-
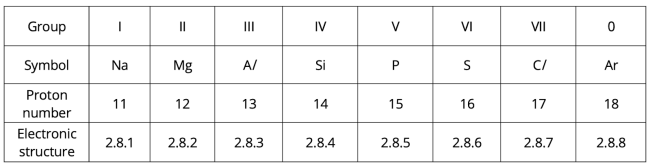
Electronic structure
The period number of the element is the same as the number of electron shells.
All elements in Period 3 have 3 electron shells (as seen in their electronic configurations).
The group number of the element is the same as the number of valence electrons.
Group I elements (e.g. sodium) have 1 valence electron. Group II elements (e.g. magnesium) have 2 valence electrons. Refer to the table above for their electronic configurations.
Hence, elements in the same group will have similar chemical properties, as they have the same number of valence electrons. One of the chemical properties shared by elements in the same group is the charge of ions.
-
Charge of ion
The group number of the elements, and hence the number of valence electrons is closely related to the number of electrons gained/lost to form ions, as well as the charge of the ions formed by the elements.
- All Group Ⅰ elements lose 1 electron to form positive ions of +1. For examples, Na+ and K+.
- All Group Ⅱ elements lose 2 electrons to form positive ions of +2. For examples, Mg2+ and Ca2+.
- All Group Ⅶ elements gain 1 electron to form negative ions of –1. For examples, F¯ and Cl¯.
- All Group 0 elements have stable electronic configurations, hence they are unreactive and they do not form compounds.
Practice Questions
Question 1:
Which property decides the order of the elements in the Periodic Table?
- the masses of their atoms
- the number of electrons in the outer shell
- the number of neutrons in the nucleus
- the number of protons in the nucleus
Solution:
(D) the number of protons in the nucleus
Explanation:
The elements in the Periodic Table are arranged in order of increasing proton number.
Question 2:
X, Y and Z are in the same period of the Periodic Table. X is a non-metal, Y is a metal, and Z shows the properties of both metals and non-metals. What is the order of these elements across the Periodic Table?
- XZY
- YXZ
- YZX
- ZXY
Solution:
(C) YZX
Explanation:
Metallic elements are found on the left-hand side of the Periodic Table. Non-metallic elements are found on the right-hand side of the Periodic Table. Those elements that show the properties of both metals and non-metals, also known as metalloids, are found between the metallic and non-metallic elements, near the zig-zag line which separates the metals from the non-metals.
Question 3:
Two elements are in the same group of the Periodic Table. Which property will be the same for both elements?
- the charge on their ions
- their electronic structure
- their melting point
- their reactivity with water or acids
Solution:
(A) the charge on their ions
Explanation:
Elements in the same group have the same number of valence electrons. Thus, they form ions of the same charge.
Question 4:
In the Periodic Table, in which period can the element of atomic number 18 be found?
- 1
- 2
- 3
- 4
Solution:
(C) 3
Explanation:
An element with atomic number of 18 has 18 protons and thus 18 electrons. Its electronic configuration is 2.8.8.
The horizontal rows in the Periodic Table are called periods. The period number indicates the number of electron shells in the element. This element has 3 electron shells, hence belong to period 3.
Question 5:
Four elements and their atomic numbers are shown in the table below.
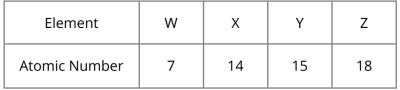
Which pair of elements is in the same group of the Periodic Table?
- W and X
- X and Y
- X and Z
- W and Y
Solution:
(D) W and Y
Explanation:
The group number indicates the number of valence electrons in the atom.
We can therefore find out the group in which these elements are by using their atomic number to figure out their electronic configuration first.
Element W: 2.5 (Group V)
Element X: 2.8.4 (Group IV)
Element Y: 2.8.5 (Group V)
Element Z: 2.8.8 (Group 0)
Hence, elements W and Y are in the same group.
Group Ⅰ - Alkali Metals
The Group Ⅰ elements are also known as alkali metals. The elements in Group I are lithium (Li), sodium (Na), potassium (K), rubidium (Rb), caesium (Cs), and francium (Fr).

Image Credits: The Photographic Periodic Table of the Elements
As these elements belong to the same group, they have similar properties.
Physical properties of alkali metals
- They are soft and can be cut by a knife easily.
- They have low melting and boiling points.
- They have low densities. Lithium, sodium, and potassium have densities lower than that of water. Hence, they float on water.
- They are good electrical and heat conductors.
- They are silvery grey in colour.
|
Image Credit: University Science Books |
Image Credit: University Science Books |
The key physical properties that can be used to identify Group I metals from the rest of the metals are the low melting and boiling points and that they are soft.
Trends in physical properties down the group
- The densities of the alkali metals increase.
- The melting and boiling points decrease.
Chemical properties of alkali metals
- They are highly reactive metals. They will readily lose 1 valence electron to form positive ions of +1 charge.
- They react spontaneously with water to form an alkali and hydrogen.
- They react with oxygen to form basic oxides.
|
Image Credit: Stanford University |
Image Credit: Science Photo Library |
Alkali metals have to be stored in oil, to prevent them from reacting with the atmospheric air (oxygen) and water.
Trend in chemical properties down the group
- The reactivity of the alkali metals increases. Lithium is the least reactive element, while francium is the most reactive element in Group I.
Why does the reactivity of the alkali metals increase down the group?
The reactivity of the alkali metal is determined by how readily its atom loses the valence electron.
As we go down a group, the size of the atom increases. The valence electron becomes further away from the nucleus. Hence, the electrostatic force of attraction between the negatively charged valence electron and the positively charged protons in the nucleus becomes weaker.
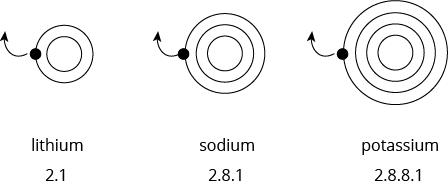
The valence electron becomes more easily lost as we go down the group. Therefore, reactivity increases down the group.
We can demonstrate the reactivity of alkali metals by their reactions with water.
Reaction with water
Group Ⅰ metals react with cold water to form an alkali and hydrogen gas.
Example:
Lithium + Water \(\xrightarrow{\qquad}\) Lithium Hydroxide + Hydrogen
2Li (s) + 2H2O (l) \(\xrightarrow{\qquad}\) 2LiOH (aq) + H2 (g)
Lithium reacts quickly with cold water. The lithium metal darts across the water surface.
Potassium + Water \(\xrightarrow{\qquad}\) Potassium Hydroxide + Hydrogen
2K (s) + 2H2O (l) \(\xrightarrow{\qquad}\) 2KOH (aq) + H2 (g)
Potassium reacts very violently with cold water. Sparks and a lilac flame are observed.
Question 6:
The element francium, Fr, is in the same group of the Periodic Table as sodium. Which of the following is most likely to be a property of francium?
- It cannot be cut by knife.
- It has poor electrical conductivity.
- It forms an ionic bromide of formula FrBr.
- It reacts mildly with water to release hydrogen.
Solution:
(C) It forms an ionic bromide of formula FrBr.
Explanation:
Sodium and francium belong to Group I of the Periodic Table, thus they form positive ions of +1 charge. As bromide ion has a charge of -1, francium and bromine react to form an ionic compound, francium bromide, with the formula FrBr.
All Group I elements are soft and can be cut by a knife easily.
As the reactivity of Group I elements increase down the group, francium will be a more reactive metal than sodium. Hence, francium will react more vigorously with water than sodium, to produce francium hydroxide and hydrogen gas.
Conclusion
In the article, we learned about the features of a Periodic Table. We understood some of the trends in the Periodic Table, as we go across a period or down a group. We also learnt about the physical and chemical properties of Group Ⅰ elements.
All these topics have been discussed keeping in mind the academic needs of Upper Secondary Chemistry students in Singapore.
Practice the examples and questions given in the article for a better understanding of the topic.
Keep Learning! Keep improving!


 SG
SG  VN
VN