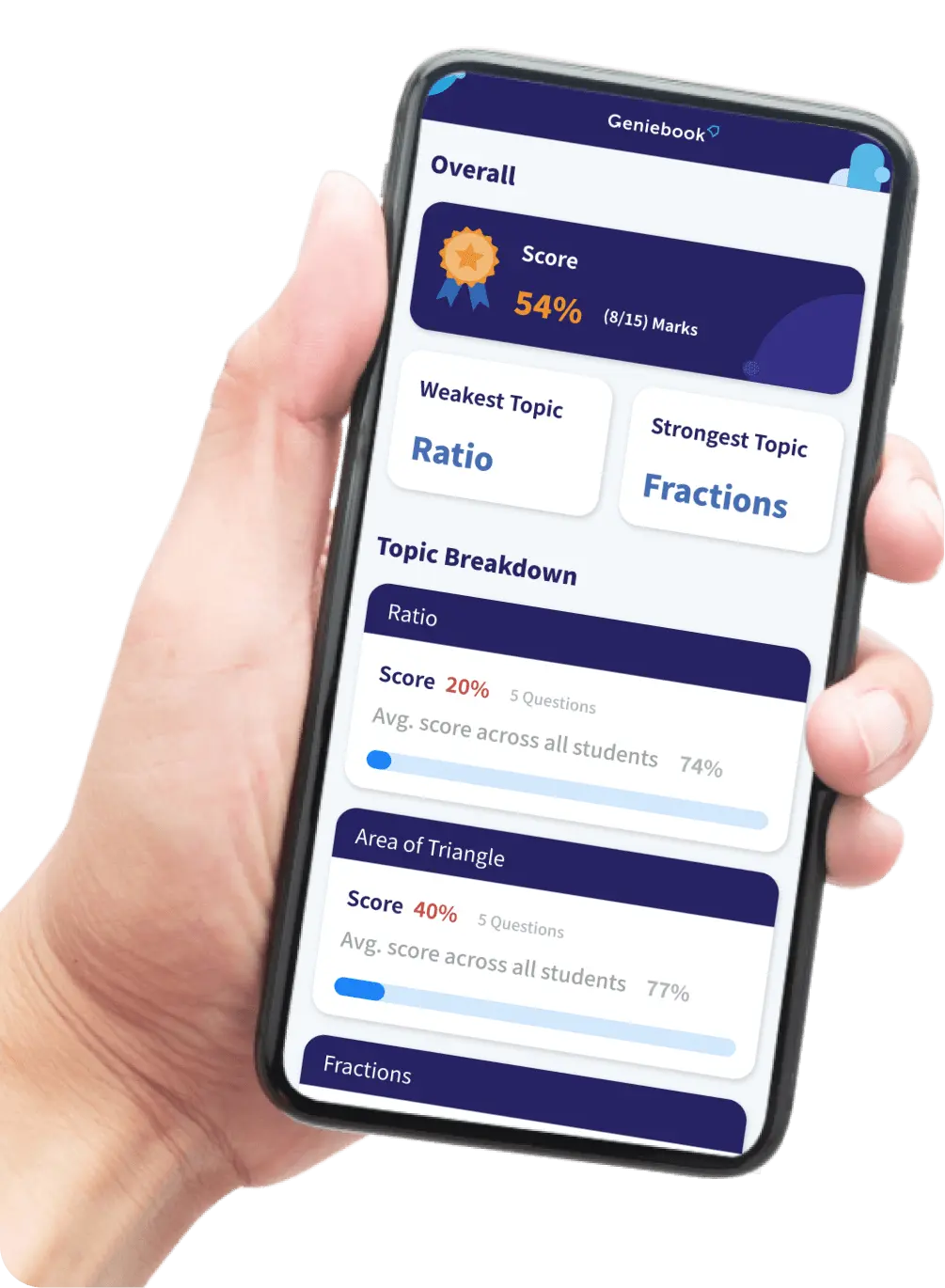
Transfer Of Thermal Energy
In this article, we are going to learn about the different ways in which thermal energy is transferred in different mediums. We will learn about the processes of conduction and convection and understand the main differences between the two processes. We will also deep-dive into how conduction and convection are used in daily life.
Thermal Energy
Thermal energy is heat.
Heat is transferred from a region of higher temperature to a region of lower temperature.
The three processes of transfer of thermal energy are:
- Conduction
- Convection
- Radiation
In this article, we will focus on conduction and convection.
Conduction
Conduction is the main process of heat transfer in solids. Conduction is the transfer of heat through a medium without any flow of the medium. In conduction, heat is transferred from one particle to another by the vibration and collisions of particles.
 |
In this picture, we see that the particles are white in colour i.e they are cold. They do not have any heat energy. |
 |
As the fire heats up the glass rod from one end, the particles gain heat and become red. As the particles vibrate and collide with the neighbouring particles, they transfer the heat energy to the neighbouring particles. |
 |
Subsequently, the heat energy is transferred to the particles at the far end of the glass rod and the whole rod becomes heated. Among solids, metals are the best conductors of heat. This is because metals contain free electrons. On top of the usual vibration and collisions of particles in conduction, metals transfer heat with an additional process of electron diffusion. Electron diffusion is the main reason why metals are good conductors of heat. |
 |
In this picture, the particles of the metal rod are white in colour indicating that they are cold and do not have thermal energy yet. The blue particles are the free electrons in metals. |
 |
As the metal rod starts heating up, the particles near the fire start gaining heat (thermal energy) and they change their colour to red. The particles transfer this heat energy to their neighbouring particles by vibration and collision. The free electrons also gain energy and they start diffusing (moving) towards the colder end of the metal rod. |
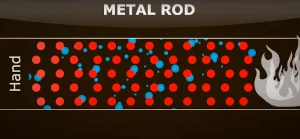 |
This movement of electrons gains more momentum as the thermal energy is gained more and more. When the free electrons have more thermal energy, they tend to collide more with the other free electrons as well as the other particles. In the process, they push the electrons having lower energy. Thus, they diffuse from one end to the other. This flow of electrons from one end of the rod to the other end is the cause of the conduction of heat in conductors. |
Practice Questions
Question 1:
The same amount of paper is wrapped around 2 rods of the same dimensions but different materials: wood and copper respectively. A Bunsen flame then licks the paper on each rod for 5 seconds each.

Image Source: GenieSmart
Which of the following is a likely observation?
- Only the paper around the wood is charred.
- Only the paper around the copper is charred.
- Papers around both wood and copper are charred.
- Papers around both wood and copper are uncharred.
Solution:
Option A is the correct answer.
Explanation:
Wood is a bad conductor of heat i.e. it does not let the heat travel through it rapidly. Thus, when the paper wound around the wood is licked by fire, the heat cannot be transferred away fast and continues to stay in the same place. Hence, as the temperature keeps increasing, it gets charred.
On the other hand, as the paper wound around the copper is heated up, it transmits that heat to the copper where the particles transmit the heat away from the fire as copper is a good conductor of heat.
Hence, only the paper wound around the wood gets charred but not the paper wound around the copper.
Question 2:
Which of the following does not explain why only paper around the wood is charred?
- Copper is a good conductor of heat.
- Wood is a good conductor of heat.
- Wood is a good insulator of heat.
- Copper conducts heat away from the paper faster than wood.
Solution:
Option B is the correct answer.
Explanation:
Copper is a good conductor of heat. It is a correct statement.
Wood is a good conductor of heat. It is a wrong statement. Since we are asked the reason that does NOT explain why only paper around the wood is charred, this option is the correct choice.
Wood is a good insulator of heat. It is a correct statement.
Copper conducts heat away from the paper faster than wood. This statement explains why the paper wound around the wood is charred and not the paper around the copper.
Do liquids conduct heat?
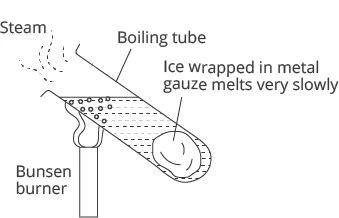
Image Source: GenieSmart
What do you think happens to the ice as the water at the top of the tube is boiling?
The ice will melt very slowly as water is a poor conductor of heat. It transmits the heat very slowly. Even though the water at the top is boiling, that heat will be transferred very slowly to the bottom. Hence, it will take a long time for the ice to melt.
Conclusion:
Liquids, especially water, are poor conductors of heat.
Do gases conduct heat?
A cooler box is shown below
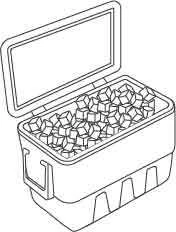
Image Source: GenieSmart
Name two features of the cooler box that help to keep its contents cool:
- The cooler box has two walls made of plastic.
- In between the two layers of plastic walls is trapped air.
Air is a poor conductor of heat. Hence, the contents in the cooler box can stay cool for a longer period of time.
Conclusion:
Gases, especially air, are poor conductors of heat.
Why are gases and liquids poor conductors/good insulators of heat?
The particles of liquids and gases are too far apart for the transfer of heat via particle vibration to be effective.
The particles in liquids and gases are too far apart that when they vibrate, they do not collide with the neighbouring particles and thus they cannot transfer the heat quickly as is done by the closely-knit particles in solids.
Hence the denser the medium, the more effective conduction is in the medium.
How then does heat transfer effectively in liquids and gases?
Convection
Convection is the transfer of heat by means of convection currents in a fluid (liquid or gas), due to a difference in its density.
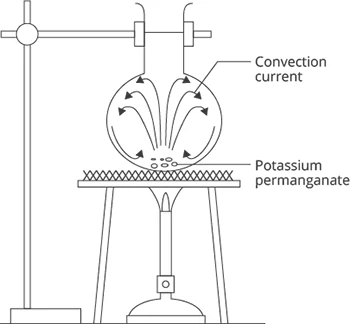
Image Source: GenieSmart
When a part of the fluid is heated, it expands (to maintain constant pressure) and its density decreases.
Warmer less dense fluid rises and cooler denser fluid sinks.
This displacement of fluid sets up a convection current.
As the fluid near the flame gets heated up, it becomes less dense and thus it rises up and is replaced by the colder, more dense fluid in its place. This process continues again and again till the whole fluid in the flask gets heated up. This is the process of convection.
What is the difference between conduction and convection?
- Heat is transferred from one particle to another without the displacement of the particles themselves. This is conduction.
- Heat is transferred through the actual displacements of the heated particles from the warmer to the cooler parts of the medium. This is convection.
Does convection occur in solids?
No. The solid particles are so tightly packed that they cannot flow/displace from one place to the other.
Question 3:
The diagram shows the cross-sectional view of a room with three positions/heights as shown.
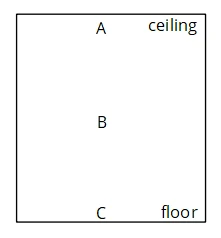
Image Source: GenieSmart
At which of the three positions should the heat radiator be placed?
- position A
- position B
- position C
Solution:
Option C is the correct answer.
Explanation:
The heat radiator should be placed on the floor at position C.
As the air becomes hot, it expands and becomes less dense. Thus, the hot air rises up and is replaced by the cold air in the upper parts of the room. Thus, the convection currents are set in motion and slowly the whole room will get warmer.
Why should the radiator be placed near the floor?
- The air surrounding the radiator at the bottom of the room is heated, expands and becomes less dense.
- Warm, less dense air rises.
- Cool, denser air sinks and replaces the warm air that has risen.
- The cool air in turn is heated up by the radiator at the bottom of the room, becomes less dense and rises again.
- This continuous displacement of air due to its density differences sets up the convection currents needed to warm up the entire room.
Question 4:
The diagram shows the cross-sectional view of a room with three positions/heights as shown.

Image Source: GenieSmart
At which of the above three positions should the air cooler be placed?
- position A
- position B
- position C
Solution:
Option A is the correct answer.
Explanation:
We place the air cooler at the top. The air surrounding the air cooler will become cold and hence will become denser. It will sink down and will be replaced by the rising hot air, which is less dense. Then this warm air will also be cooled down by the air cooler and will become denser and will sink down.
This continuous process of displacement of the cold air by the warm air will set up convection currents in the room and thus, the whole room will get cooled down.
Conclusion
In this article, we have learnt about the following topics under Thermal Transfer of Energy:
- Conduction
- Convection
- Applications in daily life
We have covered these topics as per the guidelines of the Secondary 3 Physics level.
| Continue Learning | |
|---|---|
| Light - Reflection | Units and Measurements |
| Kinematics - Distance & Displacement | Dynamics |
| Mass, Weight & Density | Thermal Properties Of Matter |
| Moments | Temperature |
| Kinetic Model of Matter | Waves |
| Work, Power & Energy | Pressure |
| Transfer Of Thermal Energy | |


 SG
SG  VN
VN