Kinetic Particle Theory
In this article, the following learning outcomes will be covered:
- Describe the properties of solid, liquid, and gaseous states of matter.
- State the kinetic particle theory.
- Explain the interconversion of the different states of matter in terms of the kinetic particle theory and energy changes for the following processes:
- melting, and
- boiling.
Properties of Matter
Properties of solid, liquid, and gas
| Property | Solid | Liquid | Gas |
|---|---|---|---|
| Shape | fixed | not fixed | not fixed |
| Volume | fixed | fixed | not fixed |
| Compressibility | cannot be compressed | cannot be compressed | can be compressed |
Kinetic Particle Theory
The kinetic particle theory states that
- all matter is made up of tiny particles, and
- these particles are in a constant and random motion.
In describing the different states of matter using kinetic particle theory, there are four properties to consider.
- Movement of particles
- Arrangement of particles
- Attractive forces between particles
- Kinetic energy of particles
Solids
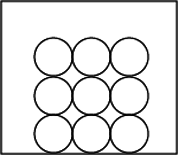
| Movement Of Particles | Vibrate and rotate about their fixed positions |
|---|---|
| Arrangement Of Particles | Very closely packed together in an orderly/regular manner |
| Attractive Forces Between Particles | Very strong |
| Kinetic Energy Of Particles | Low |
Why does a solid have a fixed shape and a fixed volume?
Due to the very strong forces of attraction between the particles of a solid, they can only vibrate and rotate about their fixed positions and hence, are unable to move about freely to take the shape of the container.
As the particles are already very closely packed together, a solid cannot be compressed.
Liquids
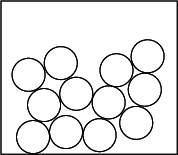
| Movement Of Particles | Slide over one another |
|---|---|
| Arrangement Of Particles | Closely packed together in a disorderly / irregular manner |
| Attractive Forces Between Particles | Strong |
| Kinetic Energy Of Particles | Moderate |
Why does a liquid not have a fixed shape?
The particles of a liquid are held by moderately strong attractive forces which prevent them from escaping from their positions. However, they can move freely by sliding over one another, allowing them to take the shape of the container.
Why does a liquid have a fixed volume?
Even though the particles of a liquid are farther away from one another than the particles of a solid, the particles of a liquid are still closely packed together with little spaces between them. Hence, the particles cannot be compressed to move even closer.
Gases
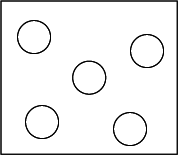
| Movement Of Particles | Move at high speeds in all directions |
|---|---|
| Arrangement Of Particles | Spaced far apart in a disorderly manner |
| Attractive Forces Between Particles | Very weak |
| Kinetic Energy Of Particles | High |
Why does a gas not have a fixed shape and a fixed volume?
Particles of a gas are held by very weak attractive forces, hence, they can move about rapidly in any direction. Therefore, a gas does not have a fixed shape.
As the particles of a gas are very far apart from each other, there are large spaces between the particles for them to move in upon the application of pressure. Since the particles can be forced to move closer together upon pressure, a gas is said to be compressible and thus has no fixed volume.
Question 1:
Which of the following statements about the kinetic particle theory is incorrect?
- It describes the different states of matter.
- It explains the changes in the state of matter.
- It explains the differences in the properties of matter.
- It explains the solubilities of different matters.
Answer:
(4) It explains the solubilities of different matters.
Question 2:
Which of the following statements about the molecules of ice is incorrect?
- The molecules have low kinetic energy.
- The molecules are held in their fixed positions.
- The molecules have weak attractive forces between them.
- The molecules vibrate and rotate about their positions.
Answer:
(3) The molecules have weak attractive forces between them.
Explanation:
Ice is in the solid state of matter. The particles of a solid are closely packed in an orderly manner, with very strong attractive forces between them. Hence, the particles can only vibrate and rotate about their fixed positions.
Question 3:
Which of the following statements provides the best example to show that matter may exist as tiny particles moving at random?
- Air rises when heated.
- Copper conducts electricity in a solid state.
- The smell of perfume can be detected in other parts of the room after the bottle is opened.
- Water cannot be compressed readily.
Answer:
(3) The smell of perfume can be detected in other parts of the room after the bottle is opened.
Explanation:
When you open a bottle of perfume, millions of tiny gas particles escape from the surface of the perfume. These tiny particles will move around freely and randomly from the bottle of perfume to other parts of the room to reach your nose, where they are detected as aromas. The spreading of aromas proves that particles of gases are constantly moving. This process by which particles move freely to fill up any available space is called diffusion.
Question 4:
Which of the following correctly describes the molecules in a very dilute sugar solution at room temperature?
| Sugar Molecules | Water Molecules | |
|---|---|---|
| 1. | Close together, moving at random | Close together, moving at random |
| 2. | Widely separated, moving at random | Close together, moving at random |
| 3. | Widely separated, moving at random | Close together, not moving |
| 4. | Widely separated, not moving | Widely separated, moving at random |
Answer:
(2) Widely separated, moving at random ----- Close together, moving at random
Explanation:
A very dilute sugar solution implies that there are fewer sugar molecules in a very large volume of water (solvent).
Since there is a large amount of water, the water molecules are closely packed together but moving at random.
The sugar molecules are dissolved in the water, which means that the particles of sugar move from a region where they are in a higher concentration (sugar lump) to where they are in a lower concentration (water). The particles of sugar move to fill up any available space between the water molecules, hence they are widely spread apart from one another, between the water molecules, and they are moving randomly.
Interconversion of States of Matter
Changes in State
Matter can change from one state to another. When matter is heated or cooled, heat energy is taken in or given out, causing the kinetic energy of the particles to change. The energy is involved in the breaking down of the attractive forces or the formation of the attractive forces between particles during changes in the physical states of a substance.
Changes of state are reversible.
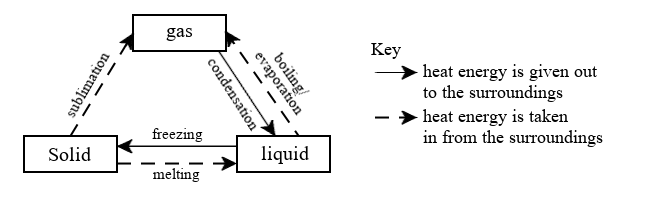
Sublimation is the process in which a solid changes directly into a gas without going through the liquid state. It occurs when heat energy is absorbed by the solid particles to turn into gas.
Freezing is the process in which a liquid changes into a solid. It takes place when heat energy is given out to the surroundings.
Melting is the process in which a solid changes into a liquid. Heat energy is absorbed from the surroundings.
Boiling is the process in which a liquid changes into a gas. In this process, heat energy is taken in from the surroundings.
Melting
Melting is a process by which a substance changes from a solid to a liquid. The temperature at which a solid becomes a liquid is known as the melting point. The melting point of a pure substance is always a constant temperature.
Changes in temperature during melting
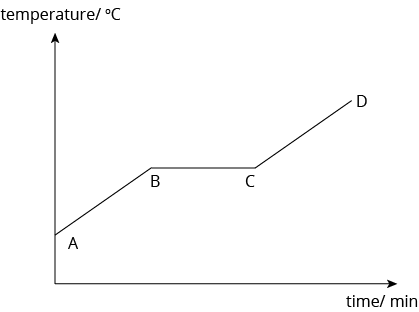
- At the region A-B, the solid is heated. The particles in the solid gain heat energy, which is converted into kinetic energy. The temperature of the solid increases until it reaches its melting point, at point B. At point B, the solid begins to melt and change into a liquid.
- At the region B-C, melting occurs. The temperature of the substance remains constant even though heating continues because the heat energy taken in by the particles is used to overcome the forces of attraction holding the particles together. Between B and C, a mixture of solid and liquid exists.
- At point C, all the solid has melted. The temperature of the liquid rises as heating continues. The liquid particles gain kinetic energy. Between C to D, only liquid exists.
Boiling
Boiling is the process by which a substance changes from a liquid to a gas. The temperature at which liquid boils is known as the boiling point. The boiling point of a pure substance is always a constant temperature.
Changes in temperature during boiling:
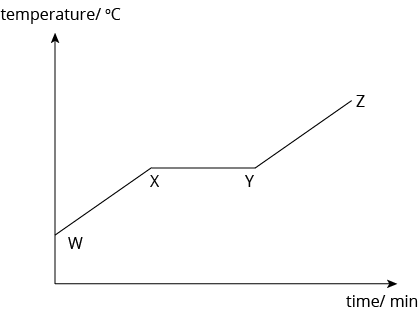
- At the region W-X, the liquid is heated, thus, the temperature of the liquid increases. The liquid particles absorb heat energy, which is converted into kinetic energy. Hence, the liquid particles start to move faster and more vigorously. The temperature of the liquid increases until it reaches the boiling point, at point X.
- At point X, boiling begins. The temperature of the substance remains constant even though heating continues because the heat energy taken in by the particles is used to overcome the forces of attraction holding the particles together. The particles will start to move further apart from one another. Between X and Y, a mixture of liquid and gas exists.
- At point Y, all the liquid has turned into a gas. The temperature continues to increase as heating continues because the gaseous particles gain heat energy which is converted into kinetic energy. Between Y to Z, only the gas exists.
Heating Curve
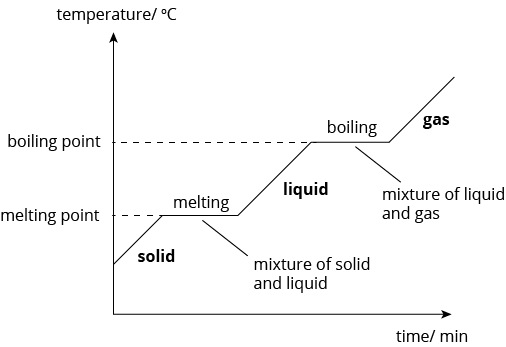
Conclusion
This article has covered different learning outcomes, from understanding the kinetic particle theory to using the kinetic particle theory to describe the states of matter and to explain the differences in the properties of solids, liquids, and gases. The kinetic particle theory is also used to explain the changes of state, in particular melting and boiling.
 SG
SG  VN
VN