Covalent Bonding
In this article, we will cover the topic of Covalent Bonding. These are the learning outcomes:
- Describe the formation of a covalent bond by the sharing of electrons.
- Draw the dot-and-cross diagram of simple covalent substances.
- Draw the structural formula of simple covalent substances.
This article is specifically written to serve the requirements for Secondary 3 Chemistry.
We have learnt in the previous topic on Ionic Bonding, that atoms of elements (except noble gases) do not have fully filled outer shells. These atoms undergo reactions to achieve the stable electronic configuration of a noble gas, either by losing outer electrons, gaining outer electrons or sharing outer electrons.
When atoms gain or lose electrons, they form ions. The metal atom loses electrons and transfer its electrons to the non-metal atoms. The positive ions (cations) and negative ions (anions) formed are attracted to each other by strong ionic bonds, forming ionic compounds.
In this article, we will learn how non-metal atoms can also react with one another to achieve the stable electronic configuration of a noble gas.
Covalent bond: The sharing of electrons
- Non-metal atoms share electrons to achieve the stable electronic configuration of a noble gas.
- A covalent bond is formed between these two atoms that share electrons with one another.
- Hence, when atoms combine by sharing electrons, molecules are formed.
- Covalent bonds can be formed between atoms of the same elements. When that happens, we get molecules of elements. For example: H2, N2, F2, O2, I2, Cl2, Br2.
- Covalent bonds can also be formed between atoms of different elements. When that happens, we get molecules of compounds. For example, H2O, CO2, CH2.
Molecules of Elements
Formation Of Hydrogen: H2
- An atom of hydrogen has one electron on its outermost shell.
- For a hydrogen atom to attain a stable duplet electronic configuration, it needs one more electron.
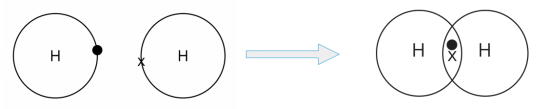
- A hydrogen atom can share its one valence electron with another hydrogen atom. In doing so, a pair of electrons is shared between the two hydrogen atoms, and a single covalent bond is formed.
We can represent the hydrogen molecule formed using either the ‘dot-and-cross’ diagram (shown on the left) or the structural formula (shown on the right).
In the structural formula, a single line between the two hydrogen atoms represents a single covalent bond.
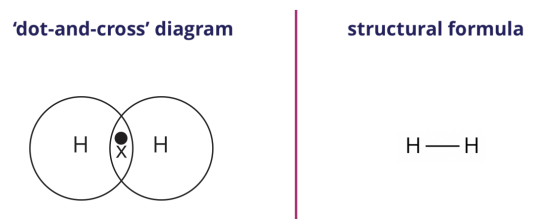
Note: When drawing the dot-and-cross diagram, use different symbols to represent electrons from different atoms.
Try It Yourself
- Draw the ‘dot-and-cross’ diagram of chlorine, Cl2. Show only the outer shell electrons.
Solution:
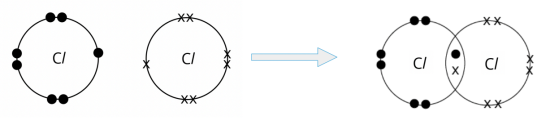
- A chlorine atom has 7 electrons on its outer shell.
- For the chlorine atom to attain a stable octet electronic configuration, it needs one more electron.
- A chlorine atom can share its one valence electron with another chlorine atom. In doing so, a pair of electrons is shared between the two chlorine atoms, and a single covalent bond is formed.
Tip: To decide on the number of valence electron(s) an atom shares in forming covalent bond(s), it is equal to the valency of the atom. Valency indicates the number of outer electrons an atom will lose or gain if it forms an ion.
For instance, a chlorine atom will need to gain one electron when it forms an ion. Thus, its valency is 1. When forming a covalent bond with another atom, such as another chlorine atom, it will share its 1 valence electron.
- Draw the structural formula of chlorine, Cl2.
Solution:
A single covalent bond is formed between the two chlorine atoms.
Hence, the structural formula of a chlorine molecule is Cl-Cl.
Formation of oxygen: O2
- An atom of oxygen has six electrons on its outermost shell.
- For an oxygen atom to attain a stable octet electronic configuration, it needs two more electrons.
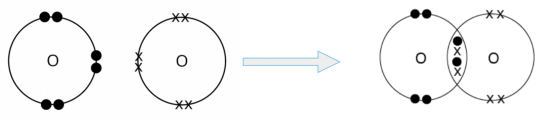
- An oxygen atom can share its two valence electrons with another oxygen atom. In doing so, two pairs of electrons are shared between the two oxygen atoms, and a double covalent bond is formed.
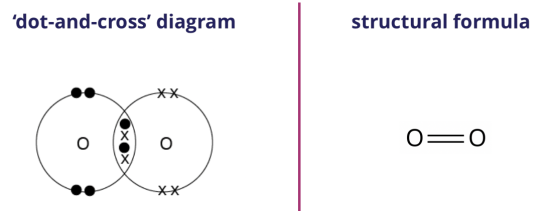
We can represent the oxygen molecule formed using either the ‘dot-and-cross’ diagram (shown on the left) or the structural formula (shown on the right).
In the structural formula, a double line ‘=’ between the two oxygen atoms represents a double covalent bond.
Formation of nitrogen: N2
- An atom of nitrogen has five electrons on its outermost shell.
- For a nitrogen atom to attain a stable octet electronic configuration, it needs three more electrons.
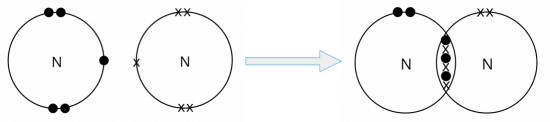
- A nitrogen atom can share its three valence electrons with another nitrogen atom. In doing so, three pairs of electrons are shared between the two nitrogen atoms, and a triple covalent bond is formed.
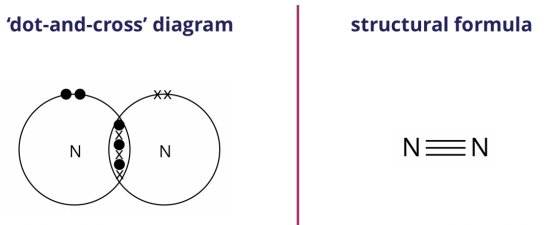
We can represent the nitrogen molecule formed using either the ‘dot-and-cross’ diagram (shown on the left) or the structural formula (shown on the right).
In the structural formula, a triple line ‘≡’ between the two nitrogen atoms represents a triple covalent bond.
Molecules of Compounds
Formation of Water: H2O
- An atom of oxygen has six electrons on its outermost shell.
- For an oxygen atom to attain a stable octet electronic configuration, it needs two more electrons.
- An atom of hydrogen has one electron on its outermost shell.
- For a hydrogen atom to attain a stable duplet electronic configuration, it needs one more electron.
- An oxygen atom can share its two valence electrons with two hydrogen atoms. In doing so, a pair of electrons is shared between the oxygen atom and each of the two hydrogen atoms. Two single O-H covalent bonds are formed.
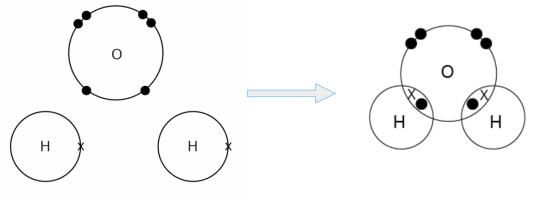
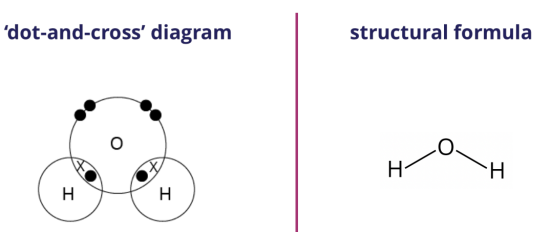
We can represent the water molecule formed using either the ‘dot-and-cross’ diagram (shown on the left) or the structural formula (shown on the right).
Formation of Carbon Dioxide: CO2
- A molecule of carbon dioxide is made up of one carbon atom and two oxygen atoms.
- An atom of oxygen has six electrons on its outermost shell.
- For an oxygen atom to attain a stable octet electronic configuration, it needs two more electrons.
- An atom of carbon has four electrons on its outermost shell.
- For a carbon atom to attain a stable octet electronic configuration, it needs four more electrons.
- A carbon atom can share its four valence electrons with two oxygen atoms. In doing so, two pairs of electrons are shared between the carbon atom and each of the two oxygen atoms. Two double C=O covalent bonds are formed.
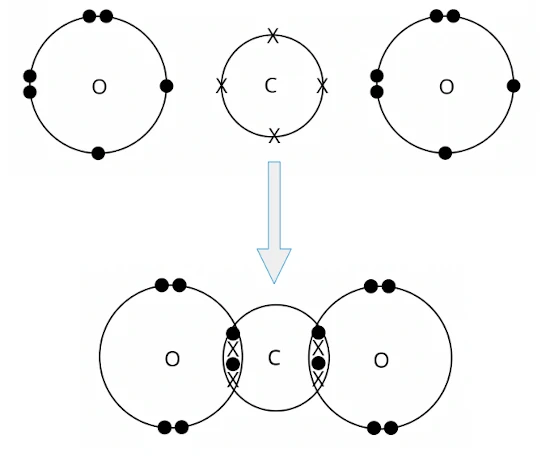
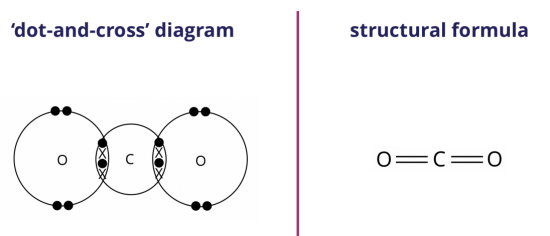
We can represent the carbon dioxide molecule formed using either the ‘dot-and-cross’ diagram (shown on the left) or the structural formula (shown on the right).
Question 1:
The table shows the electronic configurations of four elements.
| Element | Electronic Configuration |
|---|---|
| W | 1 |
| X | 2.4 |
| Y | 2.6 |
| Z | 2.8.2 |
Which of the following will form a covalent substance?
- an atom of X and two atoms of Y
- an atom of Y and an atom of Z
- an atom of Y and three atoms of W
- two atoms of X
Solution:
(A) an atom of X and two atoms of Y
Explanation:
A covalent substance is formed when two or more atoms of non-metallic element(s) are joined together by covalent bonds. Looking at the electronic configurations of the four elements, element Z is a metal, while elements W, X and Y are non-metals.
An atom of X has four electrons in its outer shell. For this atom to attain a stable octet electronic configuration, it needs four more electrons.
An atom of Y has six electrons in its outer shell. For this atom to attain a stable octet electronic configuration, it needs 2 more electrons.
Hence, an atom of X can share its four valence electrons with two atoms of Y, thus forming a covalent substance XY2.
Question 2:
What is the total number of electrons that is not being shared in a carbon dioxide molecule?
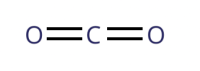
- 4
- 8
- 10
- 14
Solution:
(D) 14
Explanation:
Each oxygen atom has 8 electrons, while a carbon atom has 6 electrons.
Total number of electrons in a carbon dioxide molecule
= 8 + 8 + 6
= 22
In the structural formula of the carbon dioxide molecule, each line represents a covalent bond, or simply a pair of electrons shared between two atoms.
There are a total of 4 covalent bonds in the molecule. In other words, there are 4 pairs of shared electrons.
Total number of electrons shared
= 4 × 2
= 8
Total number of electrons not shared
= total number of electrons - total number of electrons shared
= 22 - 8
= 14
Question 3:
The following compound is made up of hydrogen and elements X, Y, and Z.
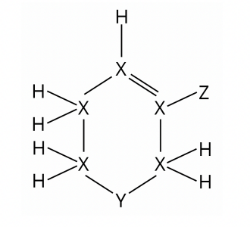
Which of the following groups of the Periodic Table are these elements most likely to be found in?
| X | Y | Z | |
|---|---|---|---|
| 1) | Group II | Group II | Group I |
| 2) | Group IV | Group II | Group I |
| 3) | Group VI | Group VI | Group VII |
| 4) | Group IV | Group VI | Group VII |
Solution:
(4)
Explanation:
The number of valence electron(s) an atom shares in forming covalent bond(s) is equal to the valency of the atom. Valency indicates the number of outer electrons an atom will lose or gain if it forms an ion.
From the structural formula, an atom of X forms 4 covalent bonds with its neighbouring atoms, by sharing its four valence electrons. Hence, the valency of X is 4. X belongs to Group IV.
An atom of Y forms 2 covalent bonds with its neighbouring atoms, by sharing its two valence electrons. Hence, the valency of Y is 2. Y belongs to Group VI.
An atom of Z forms only 1 covalent bond with its neighbouring atom, by sharing one of its valence electrons. Hence, the valency of Z is 1. Z belongs to Group VII.
Conclusion
In the article, we learned about Covalent bonds. We understood the concept of how covalent bonds are formed by the sharing of electrons between two atoms. We also learned how to represent a covalent substance using dot-and-cross diagram and structural formula. The topic of covalent bonds has been written keeping in mind the syllabus of Secondary 3 Chemistry students. Practise the examples and questions given in the article to get a better understanding of the concepts.
Keep Learning! Keep Improving.
 SG
SG  VN
VN