Ionic Bonding
In this article we will cover the topic of Ionic Bonding. These are the learning outcomes:
- Describe the stable electronic configuration of a noble gas.
- Describe the formation of ions by the gain/loss of electrons.
This article is specifically written to serve the requirements for Secondary 3 Chemistry students in Singapore.
Noble Gas
What are noble gases?
- Noble gases are elements found in Group 0 in the Periodic Table.
- The noble gases are Helium (He), Argon (Ar), Krypton (Kr), Xenon (Xe), and Radon (Rn).
- Noble gases are monatomic, which means they exist as single atoms.
- They are stable and chemically unreactive. They do not react with other elements or form compounds.
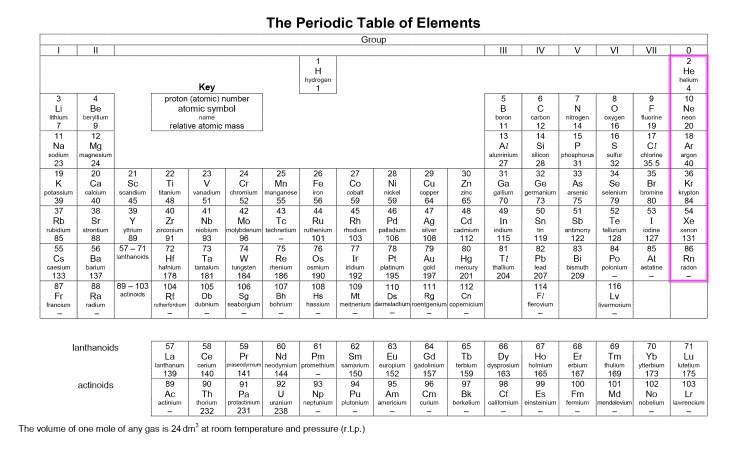
Image Credit: SEAB - seab.gov.sg
Why are noble gases unreactive?
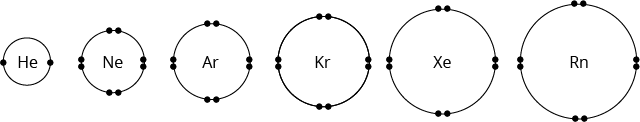
Noble gases are stable and unreactive because they have fully filled outer shells, of either a duplet or octet electronic configuration.
Helium has a duplet configuration. Its atom has a maximum of 2 electrons in the outer shell.
The other noble gases have octet configuration. Their atoms have a maximum of 8 electrons in the outer shell.
How about the other elements in the Periodic Table?
- Atoms of most other elements are reactive as they do not have fully filled outer shells.
- Hence atoms of these elements undergo reactions to gain, lose, or share their outer electrons to achieve the stable electronic configuration of noble gases.
Ions
What is an ion?
- Ions are charged particles formed when atoms lose or gain electrons. When an atom gains or loses electrons, the number of protons will not be equal to the number of electrons anymore. Hence, the atom will no longer be electrically neutral.
- Ions can be positively or negatively charged.
- Positively charged ions are called cations.
- Negatively charged ions are called anions.
- Metals form positively charged ions while non-metals form negatively charged ions.
Formation of Cations
How are cations formed?
Atoms of metals lose electrons to form cations to achieve the stable electronic configuration of a noble gas.
From sodium atom to sodium ion:
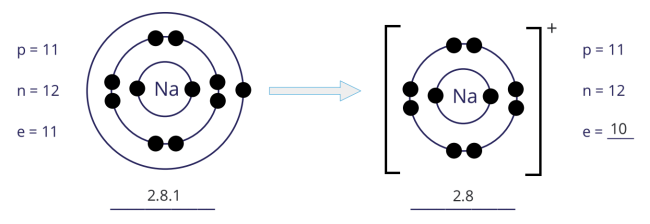
- An atom of sodium has 1 electron in its outer shell. Hence, the sodium atom loses 1 electron to achieve a stable noble gas configuration, with now a fully filled outer shell.
- In losing 1 electron, the sodium atom, from having 11 electrons and an electronic configuration of 2.8.1, becomes an ion with 10 electrons and an electronic configuration of 2.8.
- As there is now 1 more proton than electrons, the sodium ion develops a 1+ charge.
Formation of magnesium (Mg2+) ion
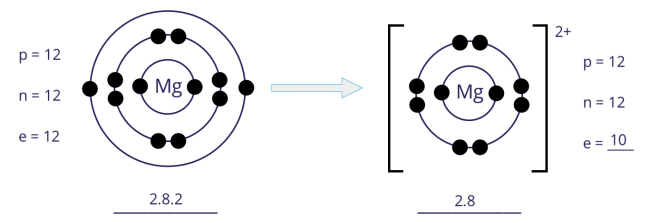
- An atom of magnesium has 2 electrons in its outer shell. Hence, the magnesium atom loses 2 electrons to achieve a stable noble gas configuration, with now a fully filled outer shell.
- In losing 2 electrons, the magnesium atom, from having 12 electrons and an electronic configuration of 2.8.2, becomes an ion with 10 electrons and an electronic configuration of 2.8.
- As there are now 2 more protons than electrons, the magnesium ion develops a 2+ charge.
Formation of aluminium (Al3+) ion
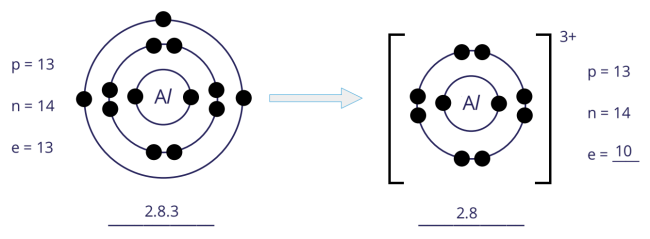
- An atom of aluminium has 3 electrons in its outer shell. Hence, the aluminium atom loses 3 electrons to achieve a stable noble gas configuration, with now a fully filled outer shell.
- In losing 3 electrons, the aluminium atom, from having 13 electrons and an electronic configuration of 2.8.3, becomes an ion with 10 electrons and an electronic configuration of 2.8.
- As there are now 3 more protons than electrons, the aluminium ion develops a 3+ charge.
Formation of Anions
How are anions formed?
Atoms of non-metals gain electrons to form anions to achieve the stable electronic configuration of a noble gas.
From fluorine atom to fluoride ion:
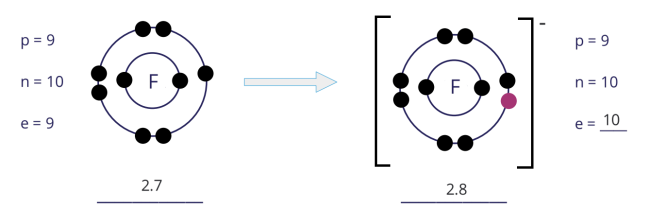
- An atom of fluorine has 7 electrons in its outer shell. Hence, the fluorine atom gains 1 electron to achieve a stable noble gas configuration, with now a fully filled outer shell.
- In gaining 1 electron, the fluorine atom, from having 9 electrons and an electronic configuration of 2.7, becomes an ion with 10 electrons and an electronic configuration of 2.8.
- As there is now 1 more electron than protons, the fluoride ion develops a 1− charge.
Formation of chloride (Cl−) ion
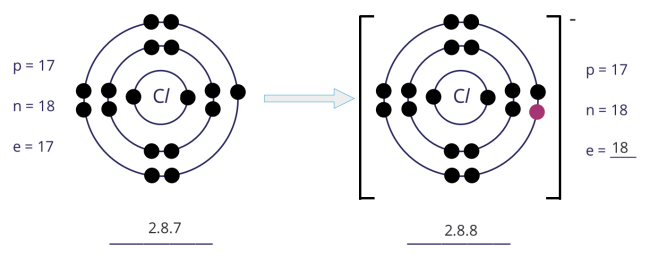
- An atom of chlorine has 7 electrons in its outer shell. Hence, the chlorine atom gains 1 electron to achieve a stable noble gas configuration, with now a fully filled outer shell.
- In gaining 1 electron, the chlorine atom, from having 17 electrons and an electronic configuration of 2.8.7, becomes an ion with 18 electrons and an electronic configuration of 2.8.8.
- As there is now 1 more electron than protons, the chloride ion develops a 1− charge.
Formation of oxide (O2−) ion
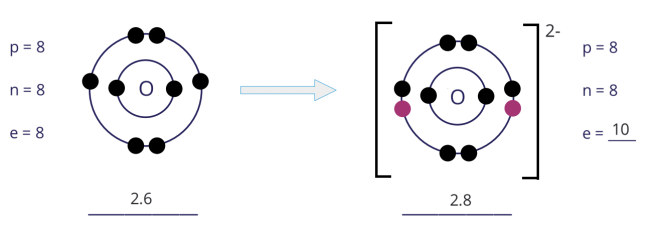
- An atom of oxygen has 6 electrons in its outer shell. Hence, the oxygen atom gains 2 electrons to achieve a stable noble gas configuration, with now a fully filled outer shell.
- In gaining 2 electrons, the oxygen atom, from having 8 electrons and an electronic configuration of 2.6, becomes an ion with 10 electrons and an electronic configuration of 2.8.
- As there are now 2 more electrons than protons, the oxide ion develops a 2– charge.
Group number & type of ions
In the Periodic Table, elements in the same group tend to form ions of the same charge.
The elements in Groups I, II, and III are metals. Their atoms lose electrons to form positive ions. The charge of the ion is the same as the group number of the element forming it.
- Elements in Group I lose 1 electron to form ions with 1+ charge. For example, Li+, Na+.
- Elements in Group II lose 2 electrons to form ions with 2+ charge. For example, Ca2+, Mg2+.
- Elements in Group III lose 3 electrons to form ions with 3+ charge. For example, Al3+.
The elements in Groups VI and VII are non-metals. Their atoms tend to gain electrons to form negative ions.
- Elements in Group VI gain 2 electrons to form ions with 2- charge. For example, O2−, S2-.
- Elements in Group VII gain 1 electron to form ions with 1- charge. For example, Br−, F-.
Other common ions and their charges
These are some other common ions that have 1+ as their charge.
| Name Of Cation | Formula Of Cation |
|---|---|
| silver | Ag+ |
| hydrogen | H+ |
These are some other common ions that have 2+ as their charge.
| Name Of Cation | Formula Of Cation |
|---|---|
| copper (II) | Cu2+ |
| zinc | Zn2+ |
| iron (II) | Fe2+ |
These are some other common ions that have 3+ as their charge.
| Name Of Cation | Formula Of Cation |
|---|---|
| chromium | Cr3+ |
| iron (III) | Fe3+ |
Polyatomic Ions
What are polyatomic ions?
Polyatomic ions are composed of two or more covalently bonded atoms, with the presence of an overall charge.
| Name Of Polyatomic Ion | Formula Of Polyatomic Ion |
|---|---|
| ammonium | NH4+ |
| hydroxide | OH– |
| nitrate | NO3– |
| carbonate | CO32– |
| sulfate | SO42– |
| phosphate | PO43– |
Conclusion
In the article, we learned about the concept of stable configuration of noble gases as well as the formation of ions by the gain or loss of electrons.
All these topics have been dealt with keeping in mind the requirements of the Secondary 3 Chemistry syllabus.
 SG
SG  VN
VN